Deposition Date
2018-07-05
Release Date
2018-09-12
Last Version Date
2024-11-20
Entry Detail
PDB ID:
6DZL
Keywords:
Title:
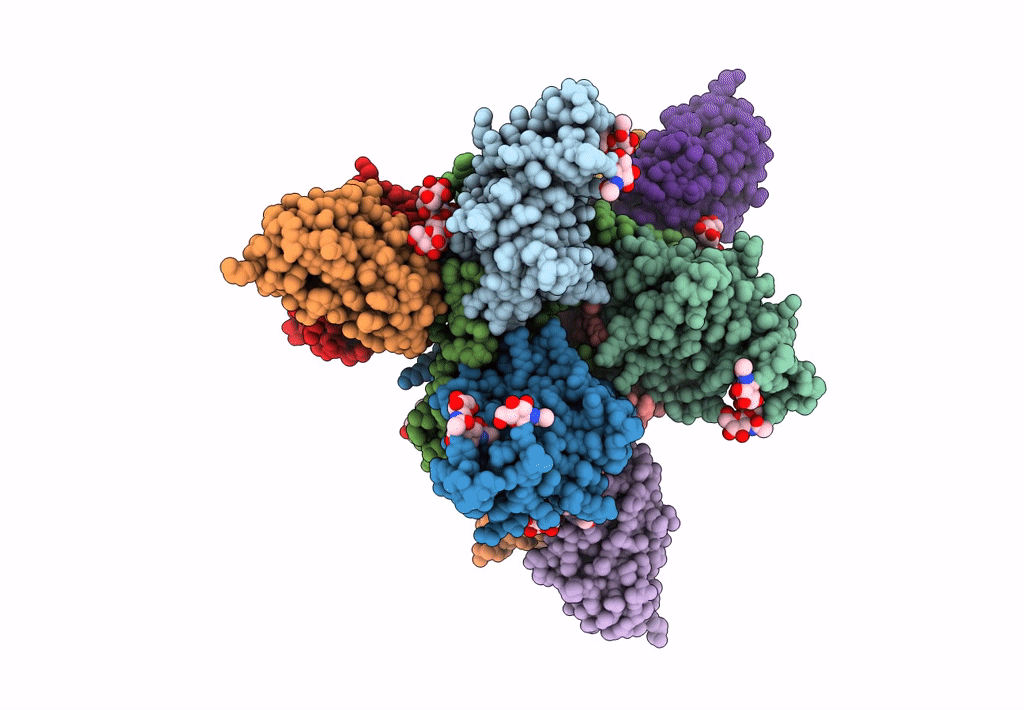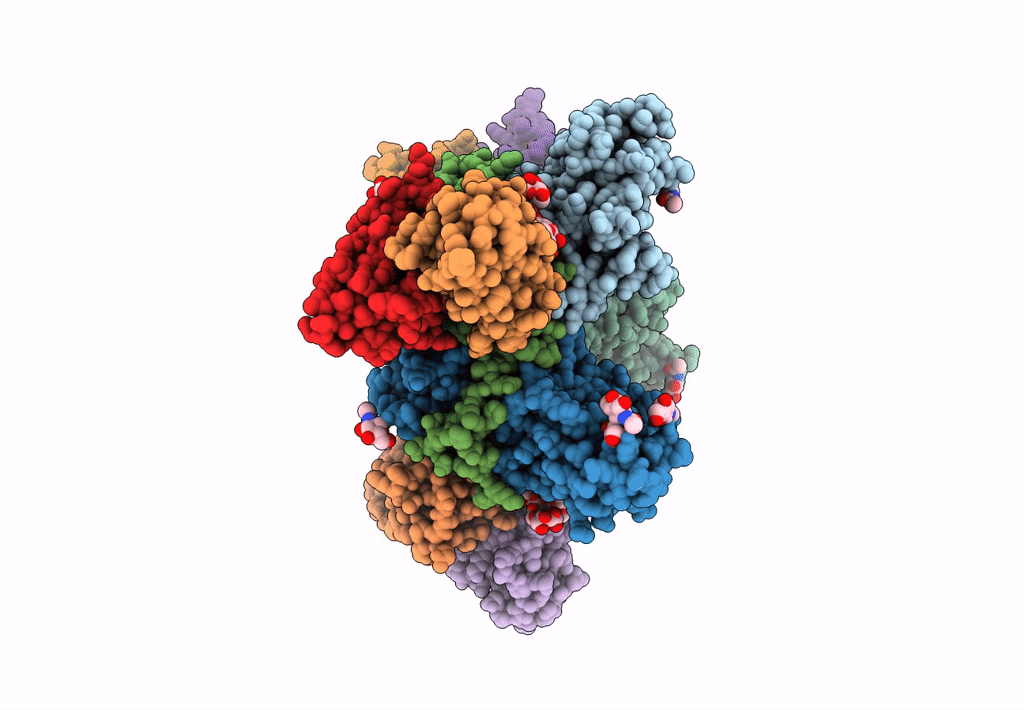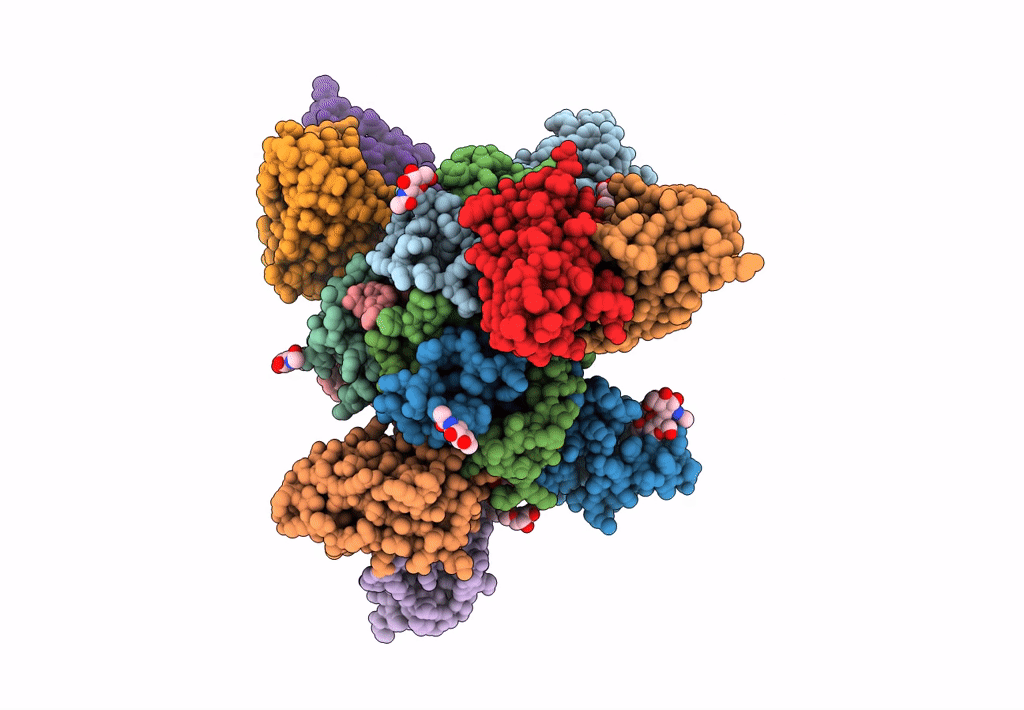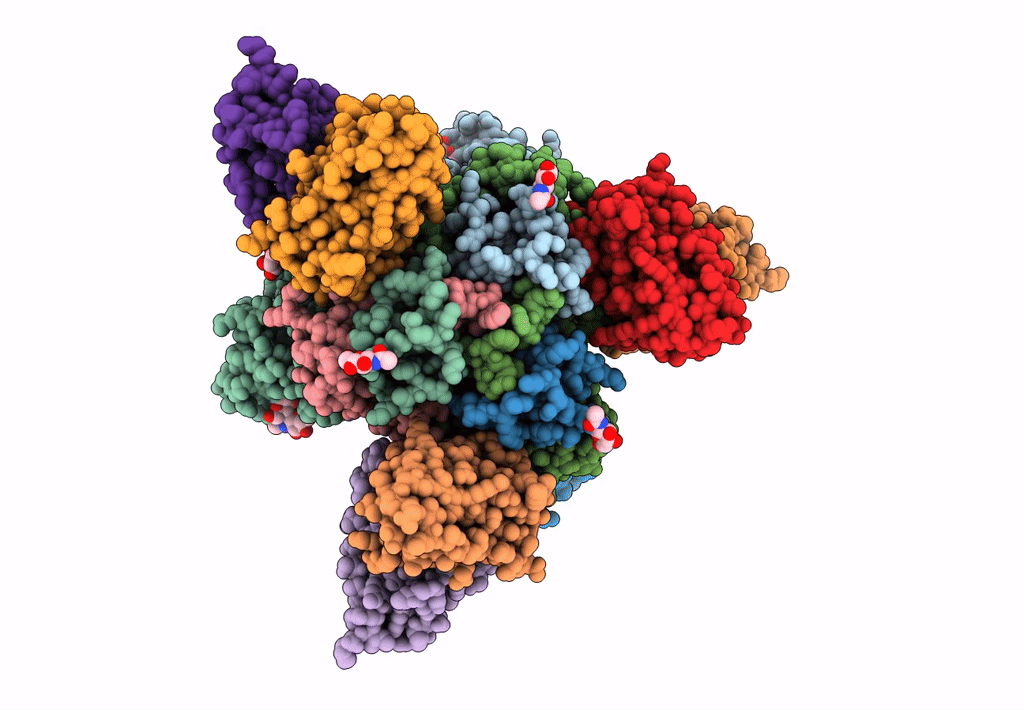
Ebola virus Makona variant GP (mucin-deleted) in complex with pan-ebolavirus human antibody ADI-15878 Fab
Biological Source:
Source Organism(s):
Zaire ebolavirus (Taxon ID: 128951)
Homo sapiens (Taxon ID: 9606)
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
4.14 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE


