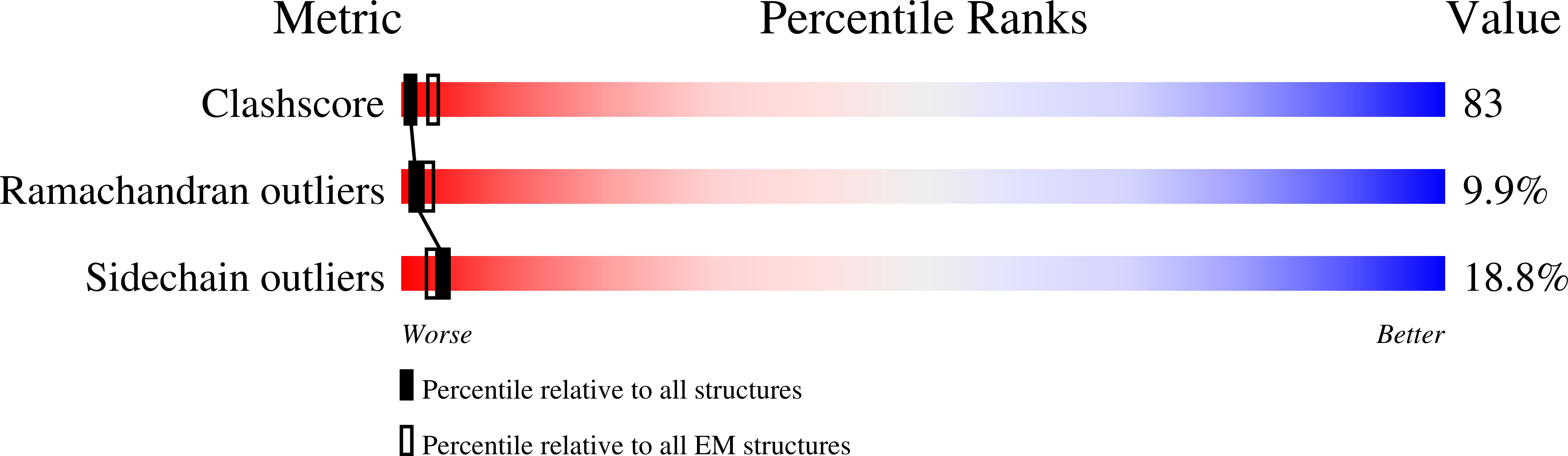
Deposition Date
2010-11-30
Release Date
2011-06-01
Last Version Date
2024-02-21
Entry Detail
PDB ID:
3IZQ
Keywords:
Title:
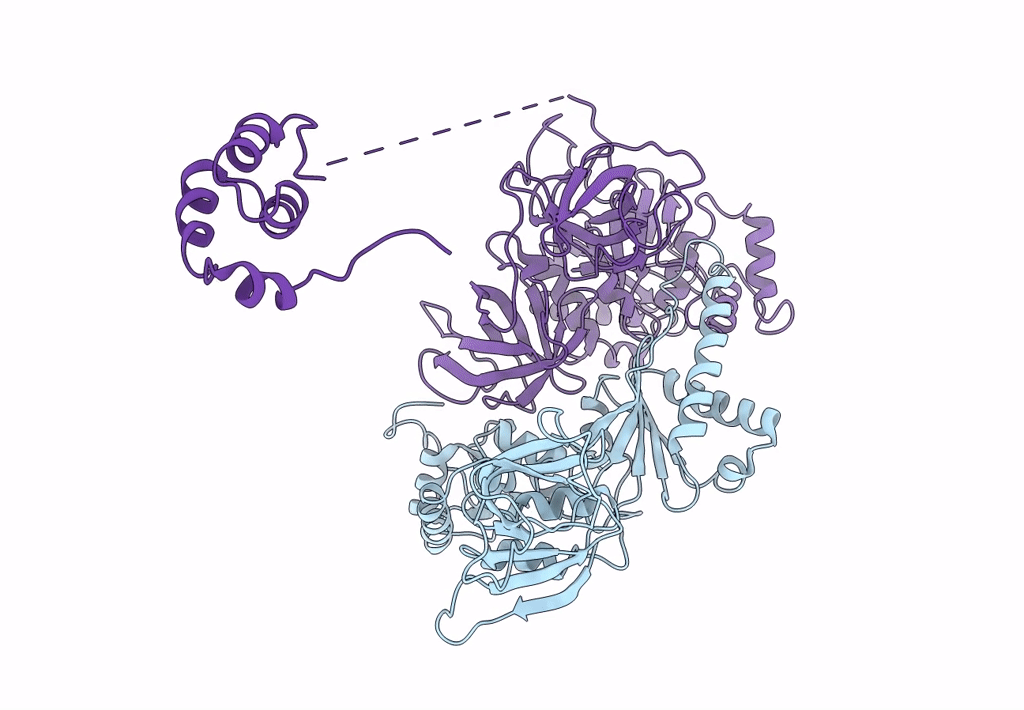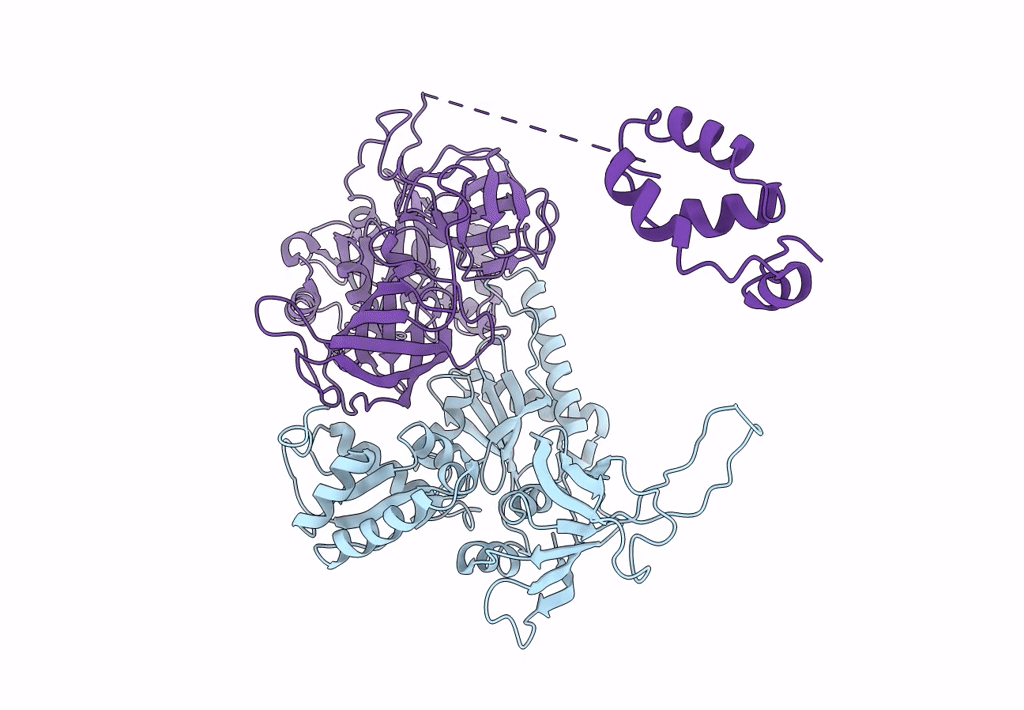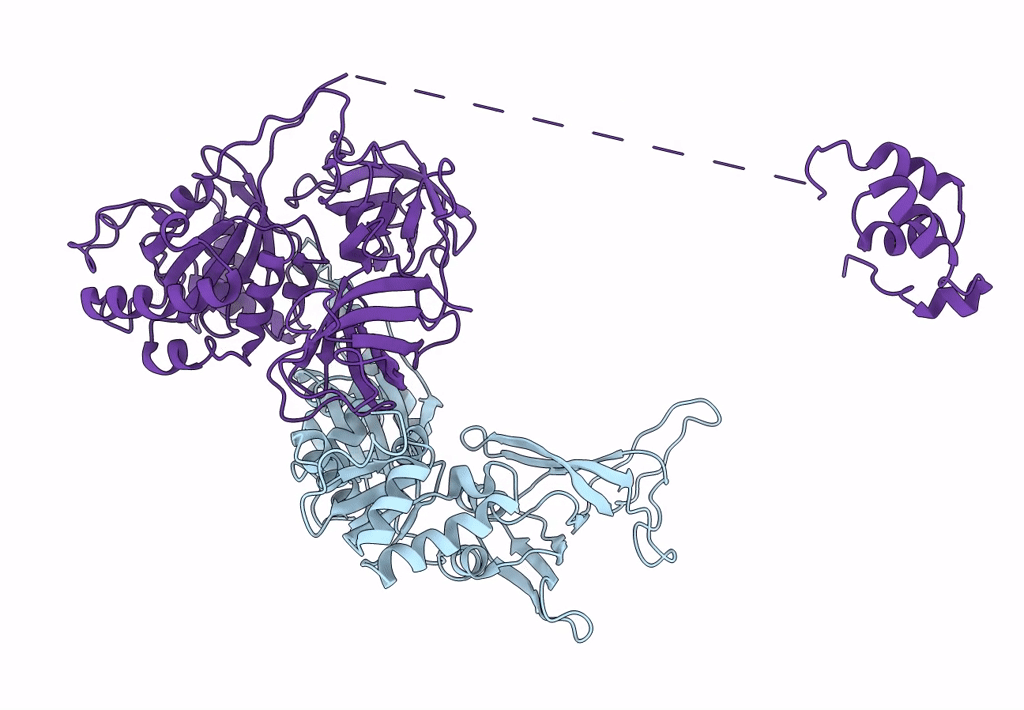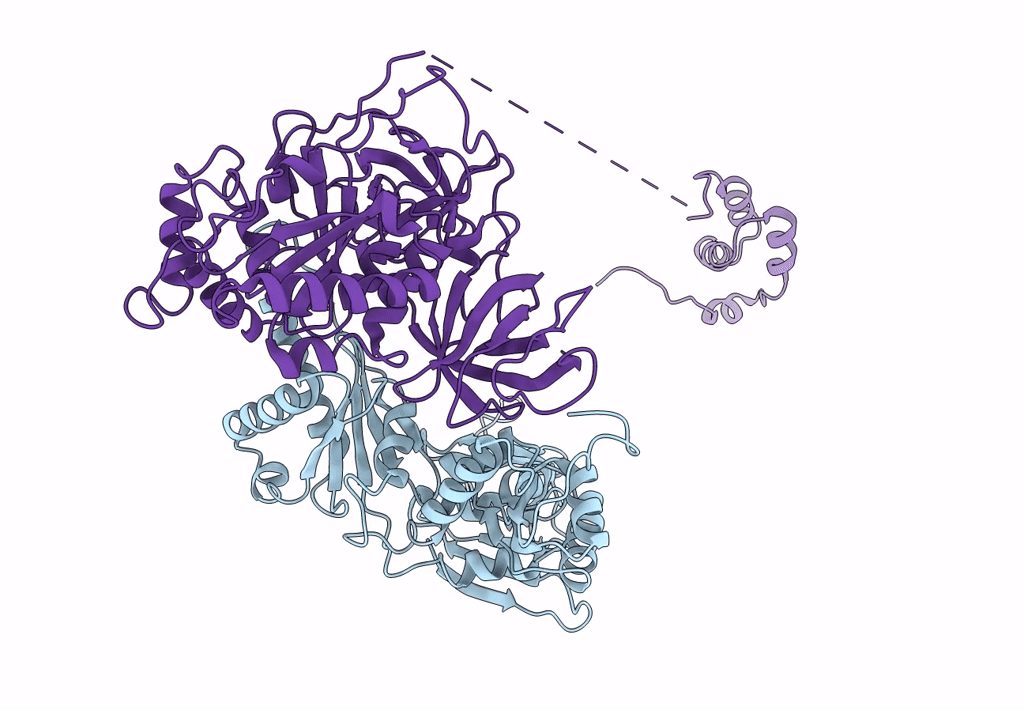
Structure of the Dom34-Hbs1-GDPNP complex bound to a translating ribosome
Biological Source:
Source Organism(s):
Saccharomyces cerevisiae (Taxon ID: 4932)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
9.50 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE