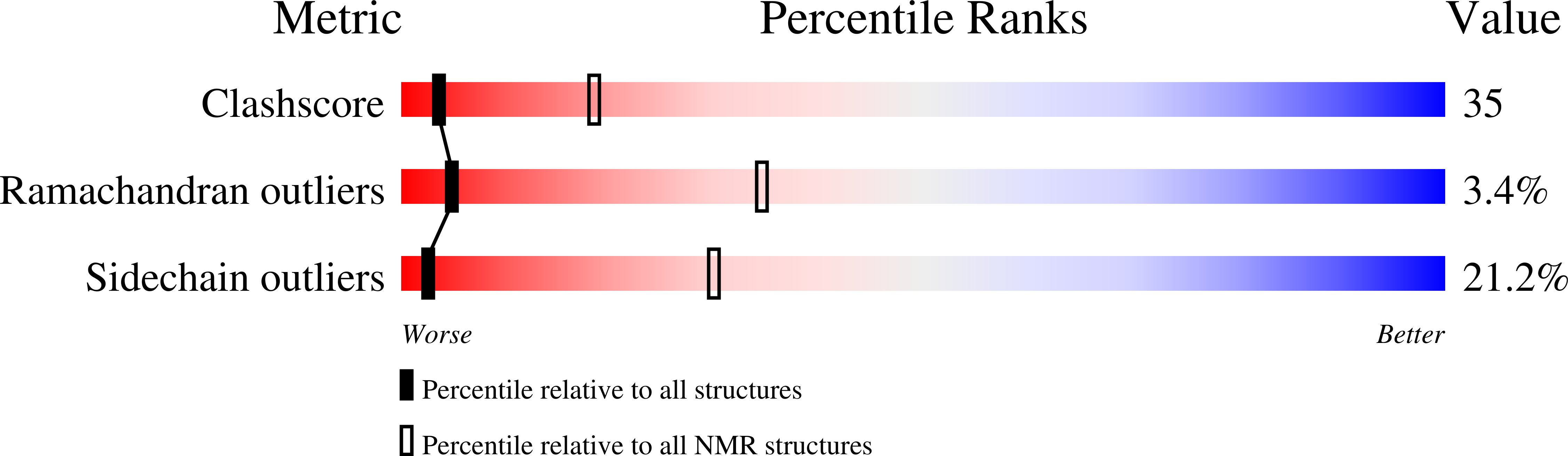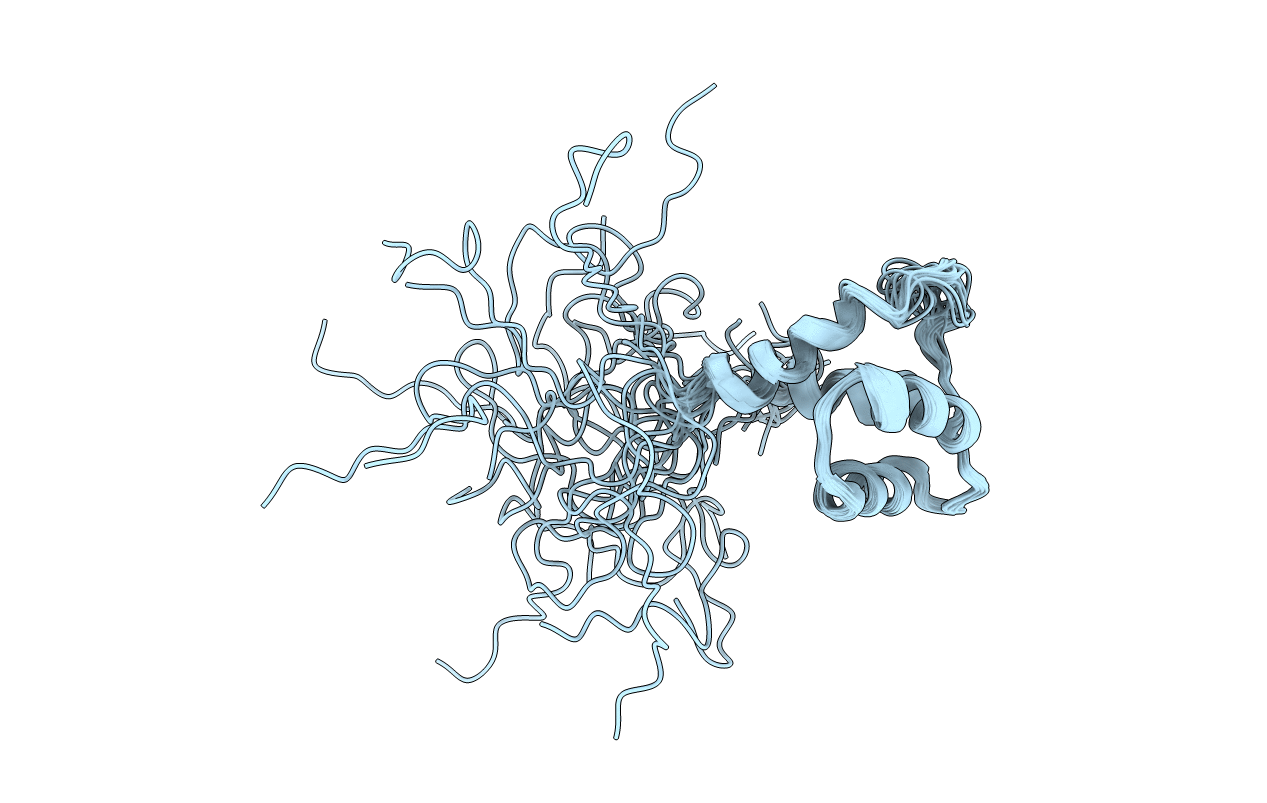
Deposition Date
2012-12-06
Release Date
2013-12-11
Last Version Date
2024-05-15
Entry Detail
PDB ID:
2M1U
Keywords:
Title:
Solution structure of the small dictyostelium discoideium myosin light chain mlcb provides insights into iq-motif recognition of class i myosin myo1b
Biological Source:
Source Organism(s):
Saccharomyces cerevisiae (Taxon ID: 4932)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
200
Conformers Submitted:
21
Selection Criteria:
structures with the lowest energy