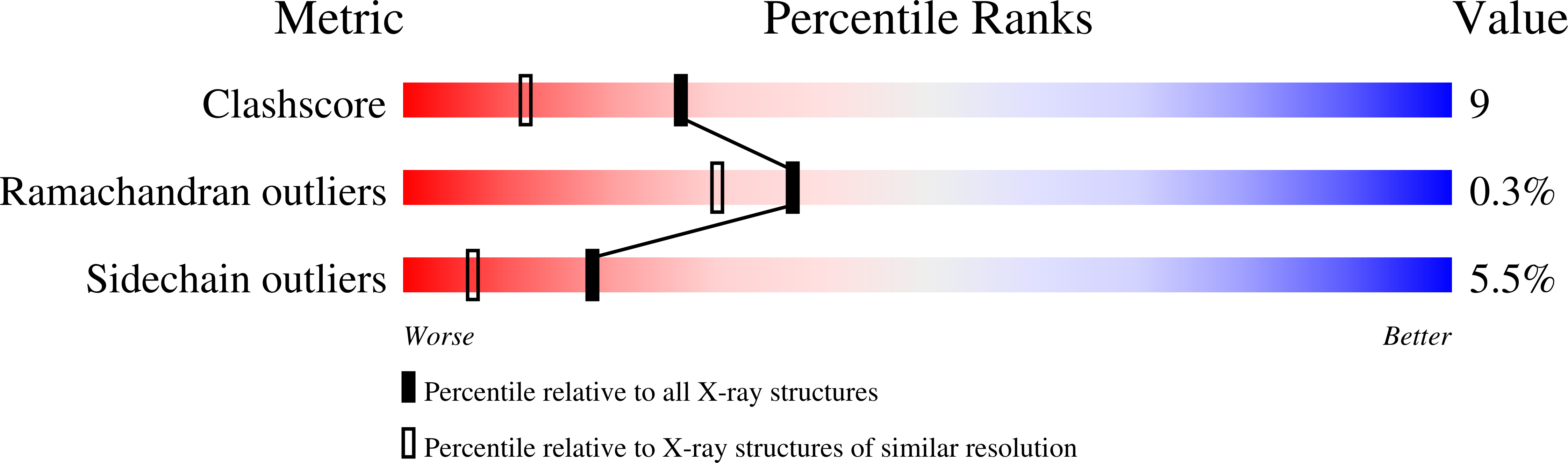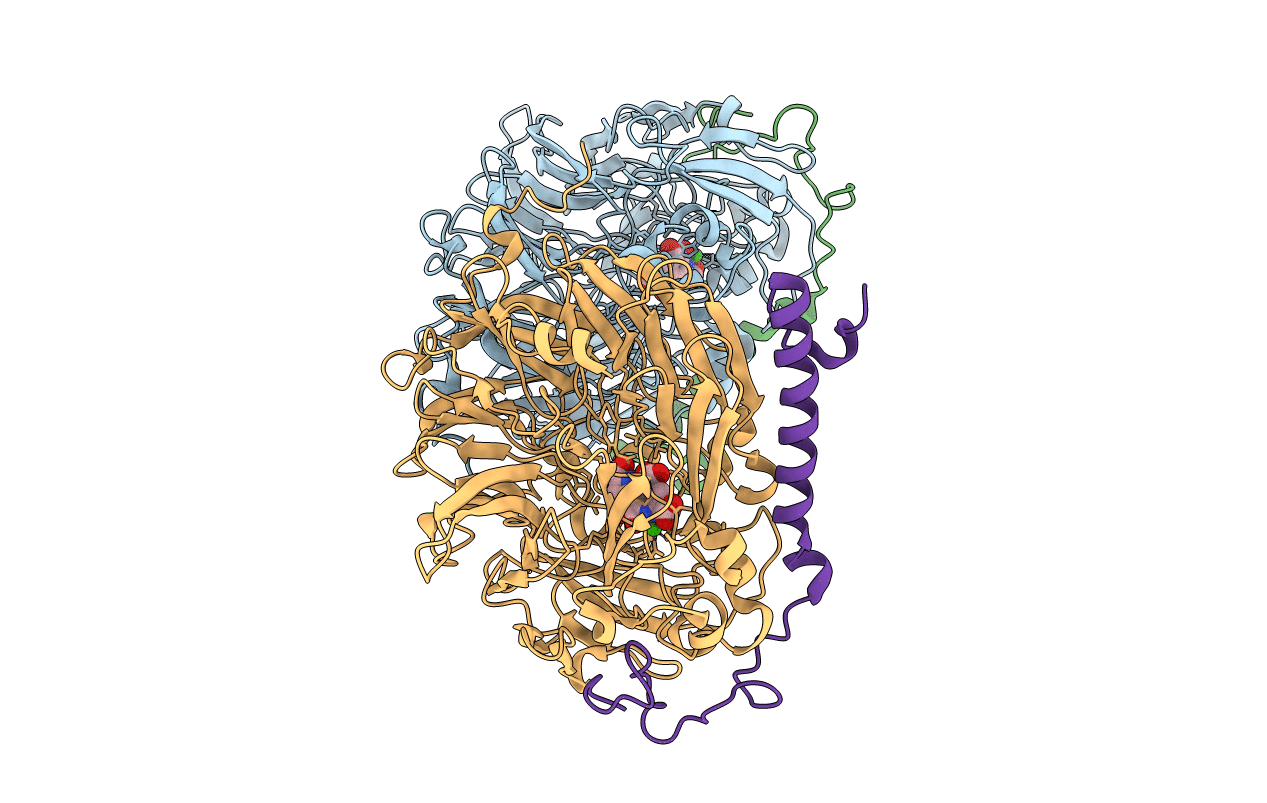
Deposition Date
2001-05-11
Release Date
2001-06-14
Last Version Date
2024-11-06
Method Details:
Experimental Method:
Resolution:
1.94 Å
R-Value Free:
0.22
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 1