Deposition Date
2025-05-13
Release Date
2026-05-06
Last Version Date
2026-05-06
Entry Detail
PDB ID:
9UXB
Keywords:
Title:
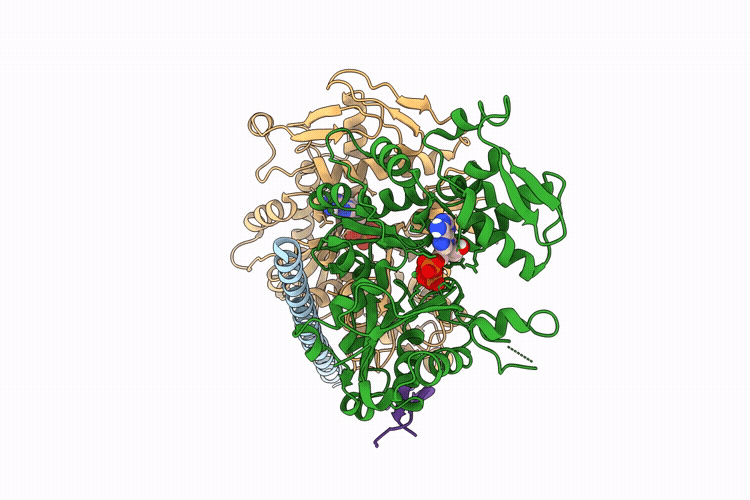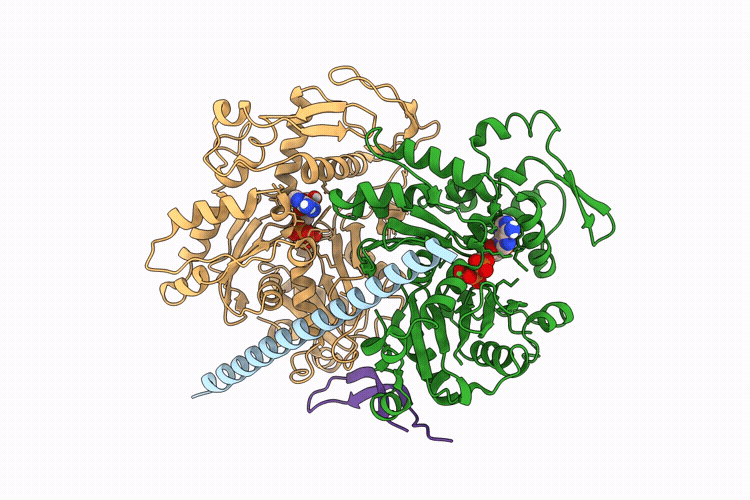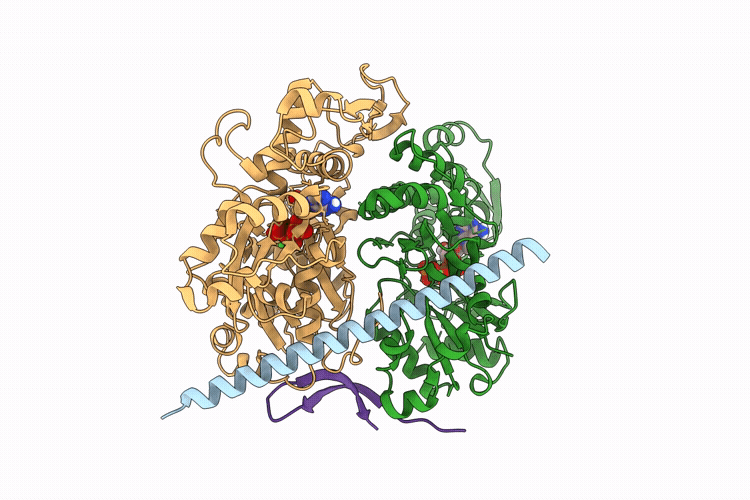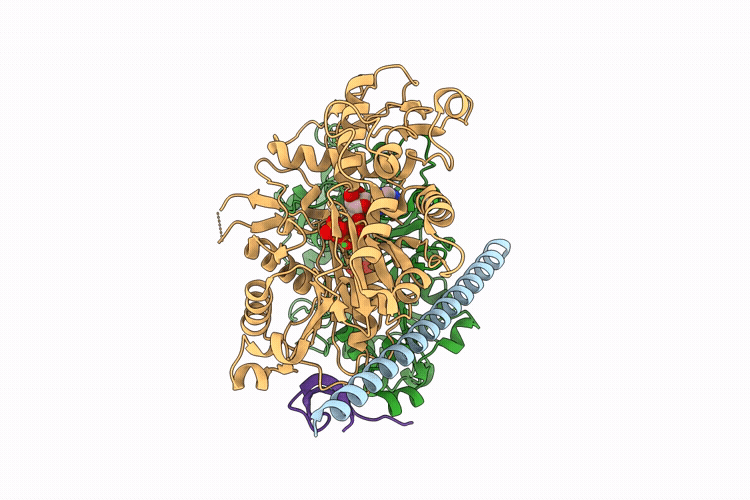
The apo structure of BCL7B-containing ARP module of the human SWI/SNF complex
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
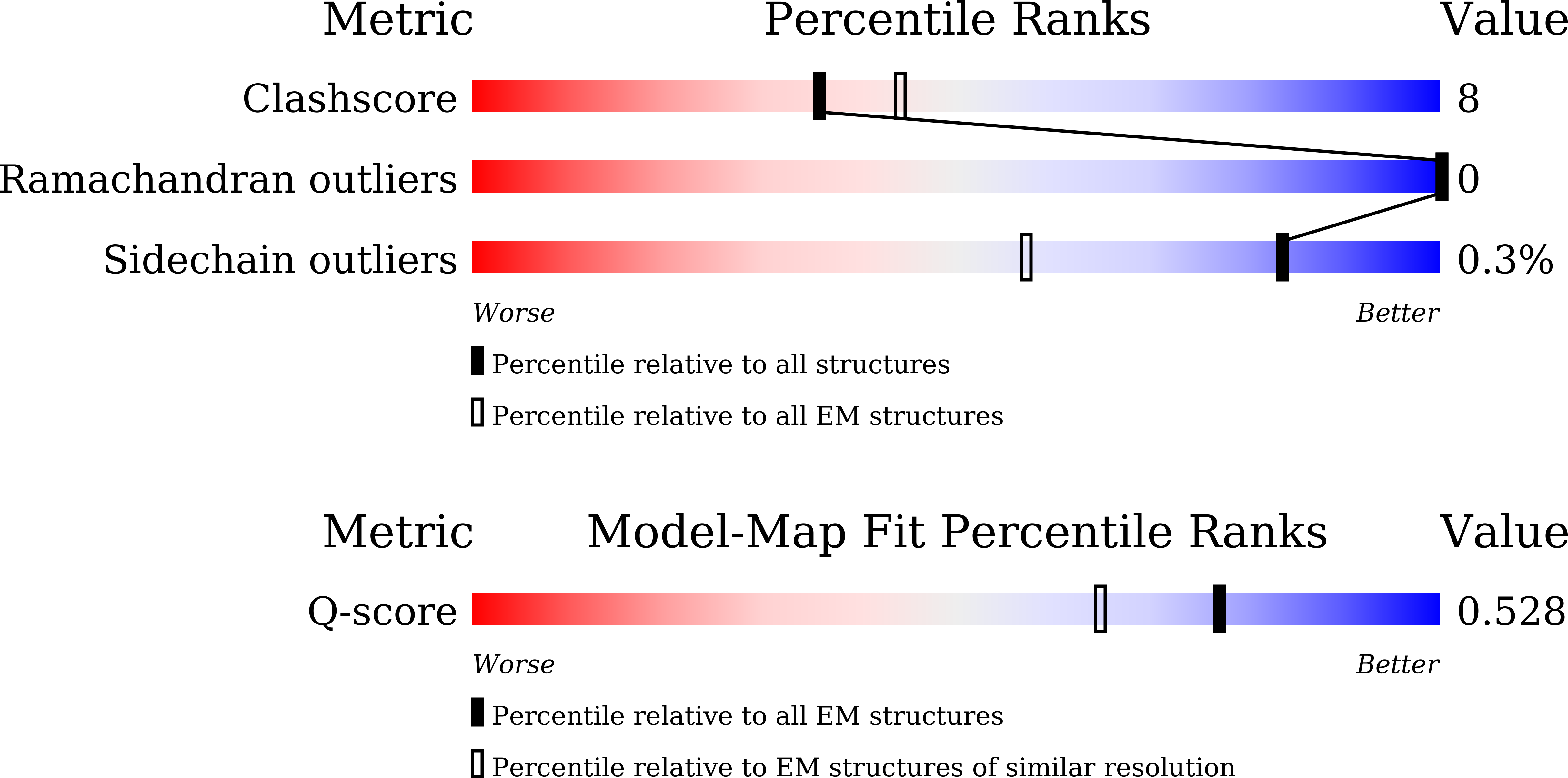
Experimental Method:
Resolution:
2.92 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE