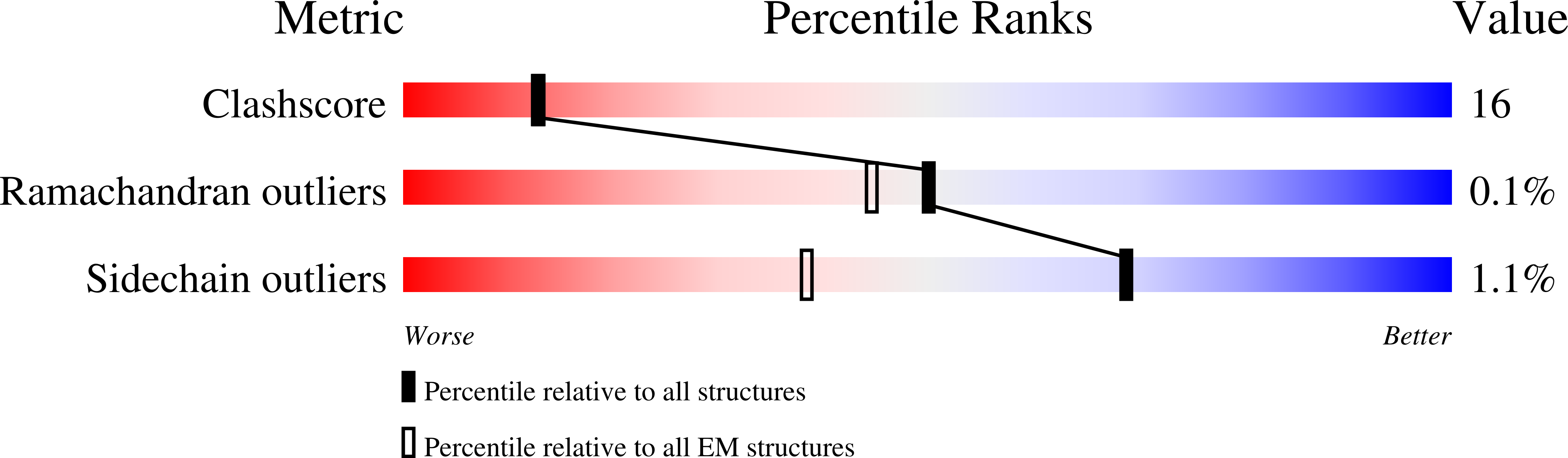
Deposition Date
2025-06-24
Release Date
2025-08-27
Last Version Date
2025-08-27
Entry Detail
PDB ID:
9RPD
Keywords:
Title:
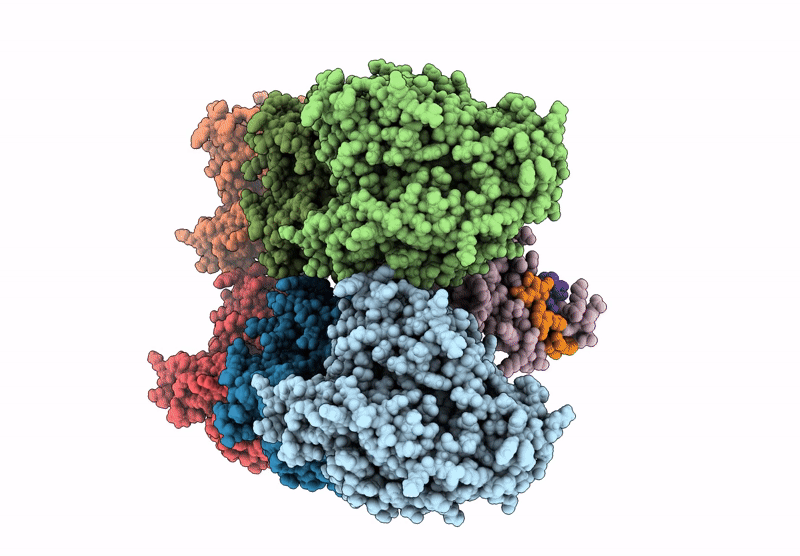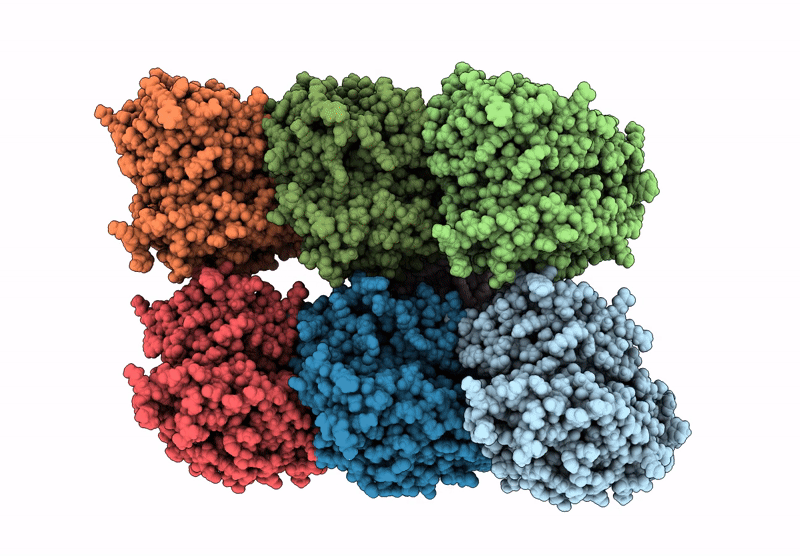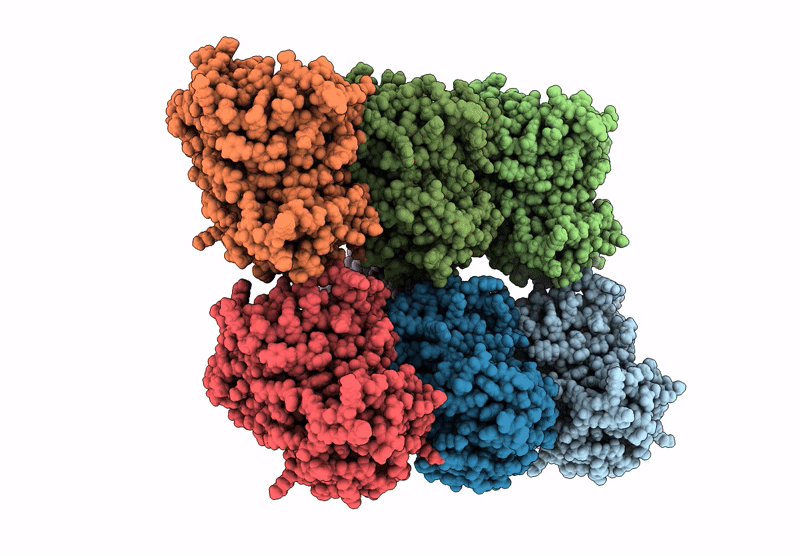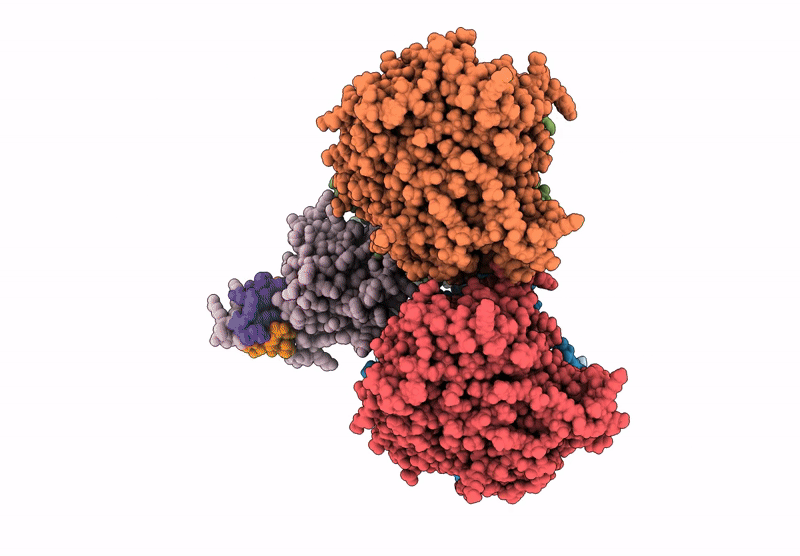
D. melanogaster Augmin TII N-clamp (GST-fusion) bound to a microtubule, well-defined subset of particles
Biological Source:
Source Organism(s):
Drosophila melanogaster (Taxon ID: 7227)
Aequorea victoria (Taxon ID: 6100)
Schistosoma japonicum (Taxon ID: 6182)
Sus scrofa (Taxon ID: 9823)
Aequorea victoria (Taxon ID: 6100)
Schistosoma japonicum (Taxon ID: 6182)
Sus scrofa (Taxon ID: 9823)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
4.90 Å
Aggregation State:
HELICAL ARRAY
Reconstruction Method:
SINGLE PARTICLE