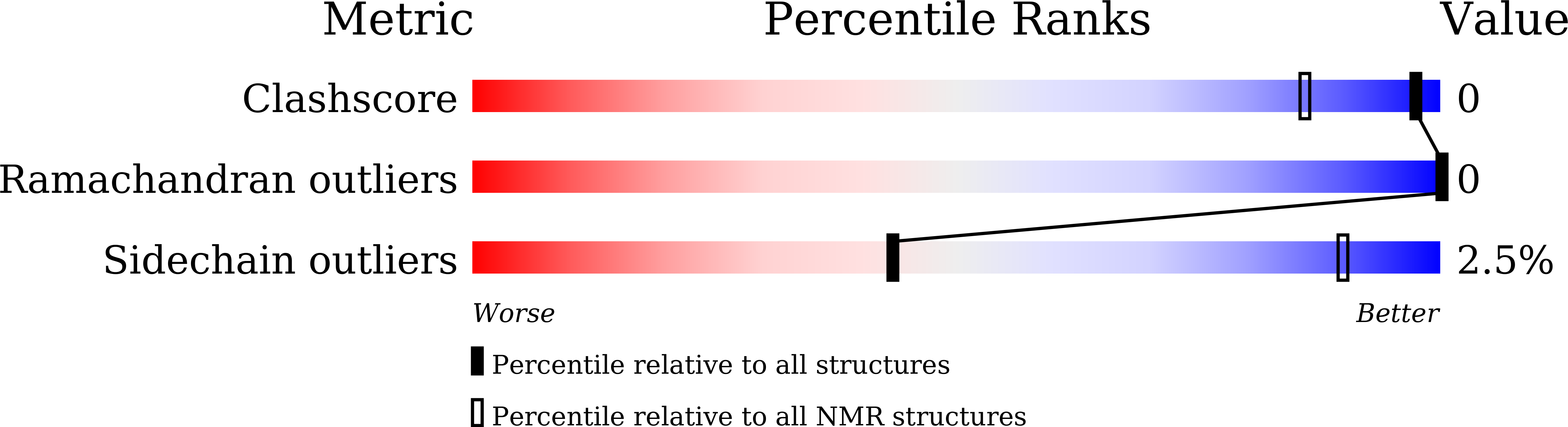
Deposition Date
2025-02-26
Release Date
2025-09-03
Last Version Date
2026-03-18
Entry Detail
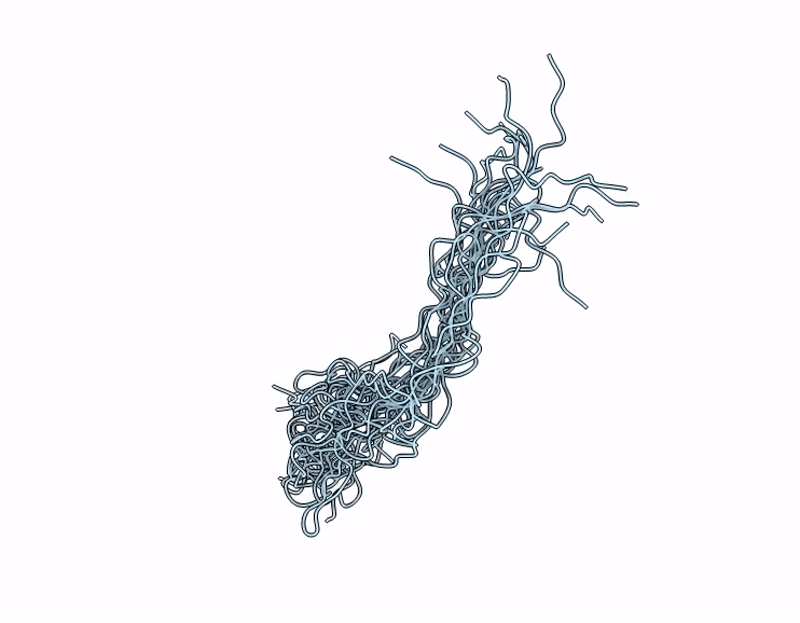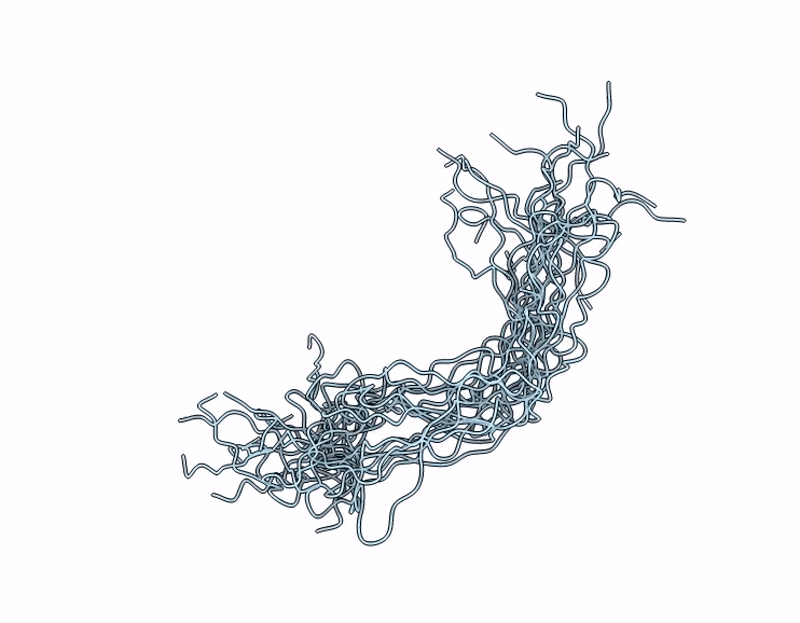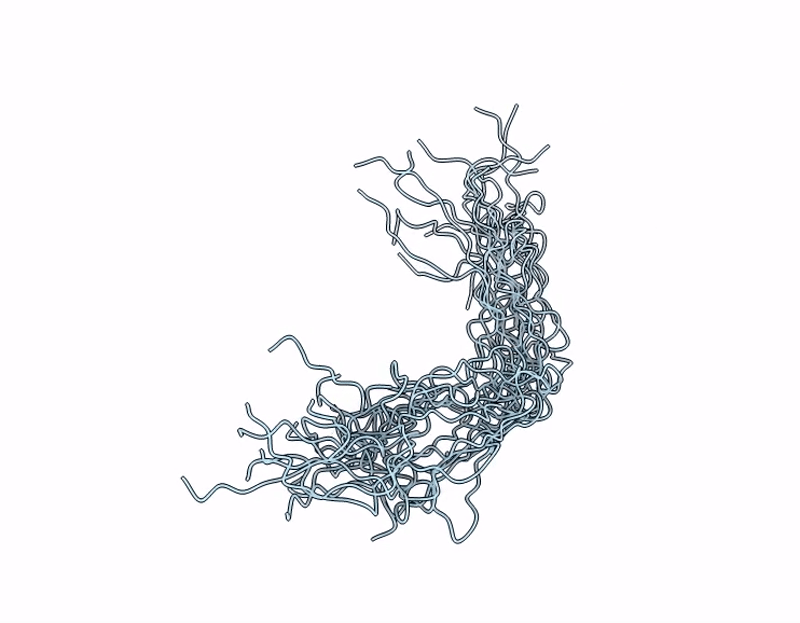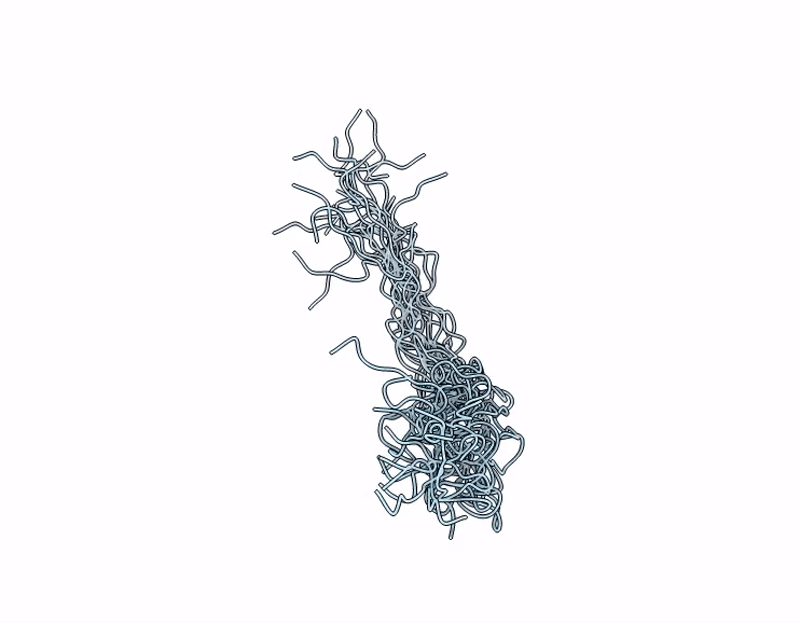
PDB ID:
9M1Y
Keywords:
Title:
NMR Structure of Mouse Keratin 17 Tail Domain (G390 - R433) in solution
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
200
Conformers Submitted:
17
Selection Criteria:
structures with the lowest energy