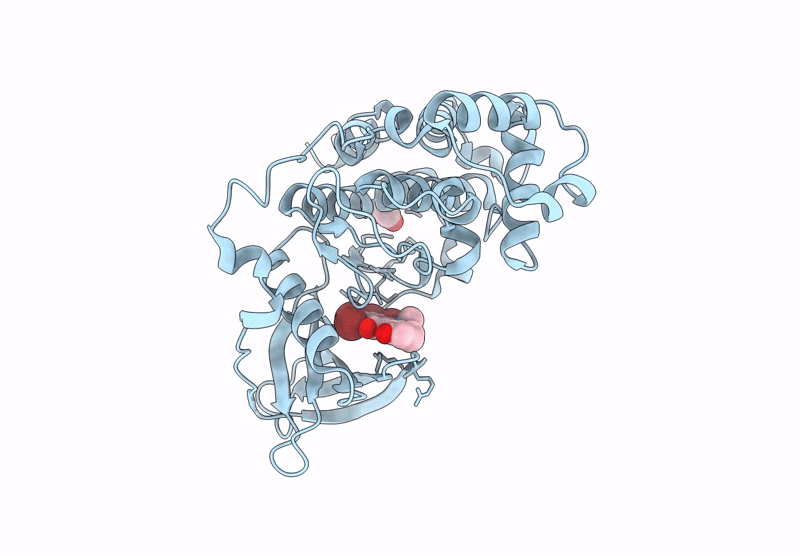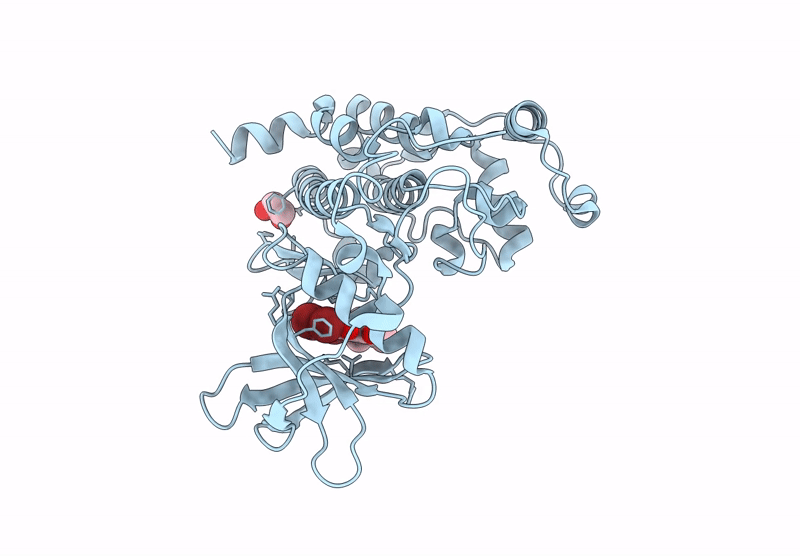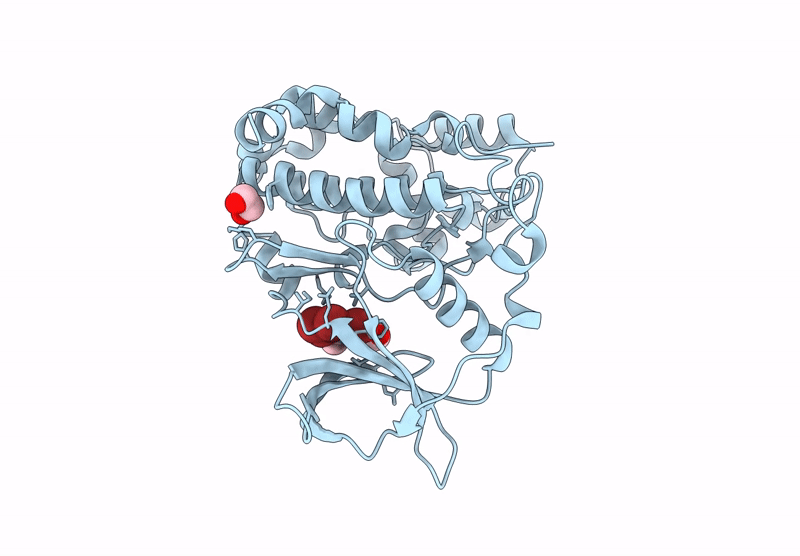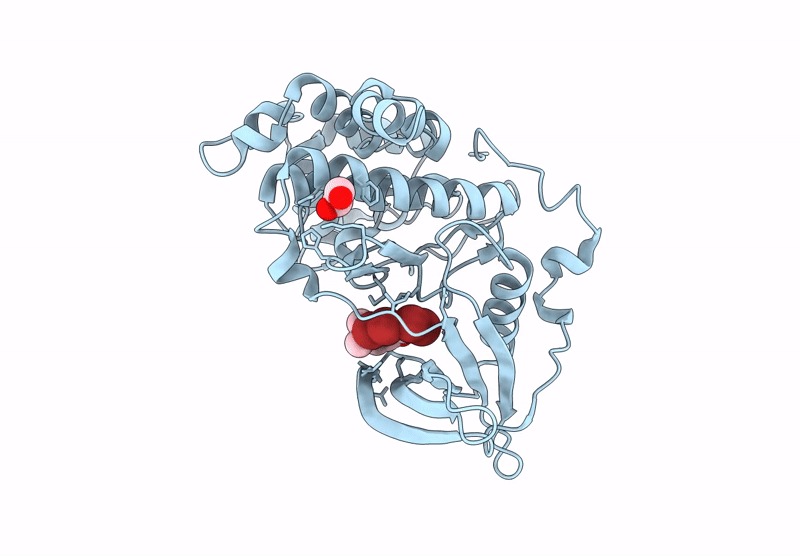
Abstact
Protein kinase CK2 is a promising therapeutic target, especially in oncology. Over the years, various inhibitors have been developed, with polyhalogenated scaffolds emerging as a particularly effective class. Halogens like bromine and chlorine enhance inhibitor stability by forming additional interactions within the ATP pocket. Among halogenated scaffolds, benzotriazole and benzimidazole have led to potent molecules such as 4,5,6,7-tetrabromo-1H-benzotriazole (IC50 = 300 nM) and 4,5,6,7-tetrabromo-2-(dimethylamino)benzimidazole (IC50 = 140 nM). Modifications, including 4,5,6-tribromo-7-ethyl-1H-benzotriazole (IC50 = 160 nM), further improved activity. Changing scaffolds while retaining halogens has enabled design of new inhibitors. Flavonols, dibenzofuranones, and the indeno[1,2-b]indole scaffold are key examples. Halogenation of the reference molecule 5-isopropyl-5,6,7,8-tetrahydroindeno[1,2-b]indole-9,10-dione (4b, IC50 = 360 nM) significantly boosted potency. The study focused on introducing four halogens, yielding to the compound 1,2,3,4-tetrabromo-5-isopropyl-5,6,7,8-tetrahydroindeno[1,2-b]indole-9,10-dione (MC11), with an IC50 of 16 nM. Co-crystallography revealed how bromine atoms enhance binding, and MC11 demonstrated strong in cellulo activity, particularly against leukemic cell lines like IPC-Bcl2.