Deposition Date
2024-08-09
Release Date
2025-08-20
Last Version Date
2026-03-18
Entry Detail
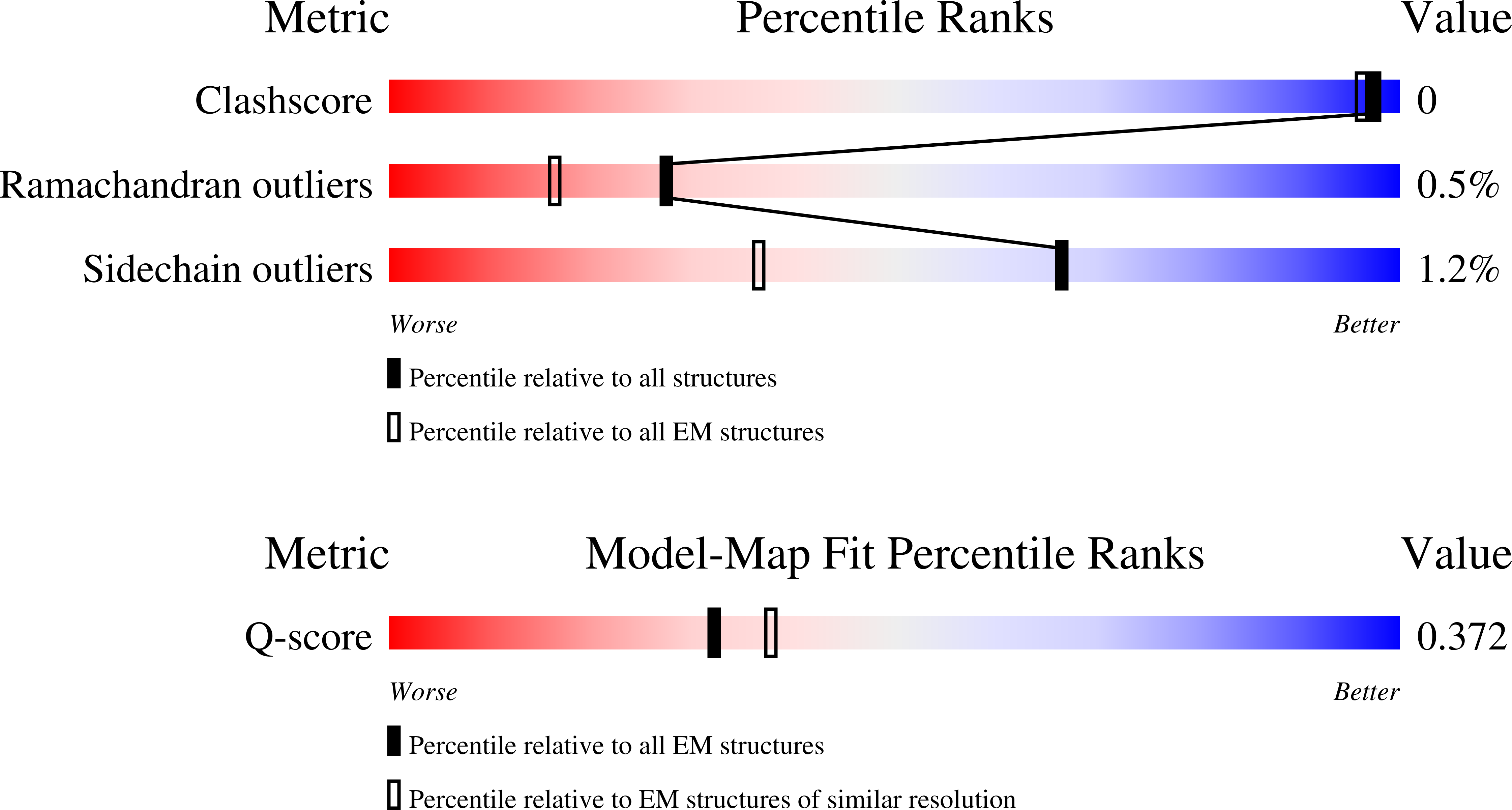
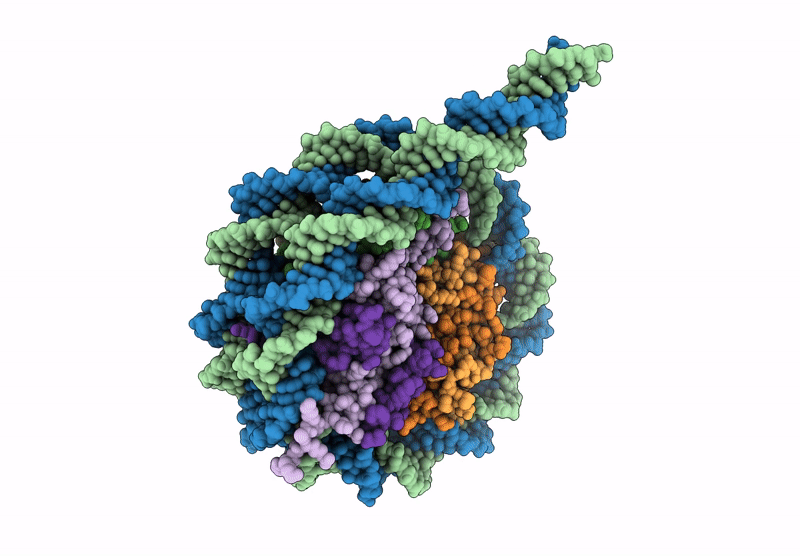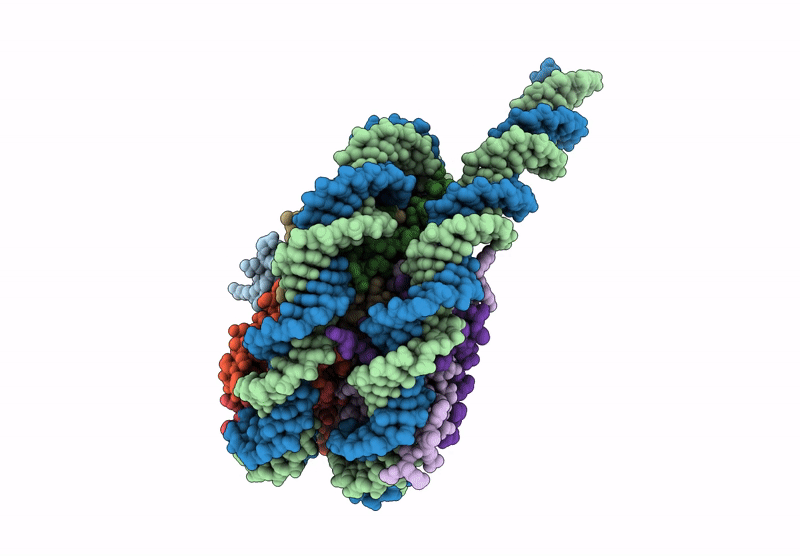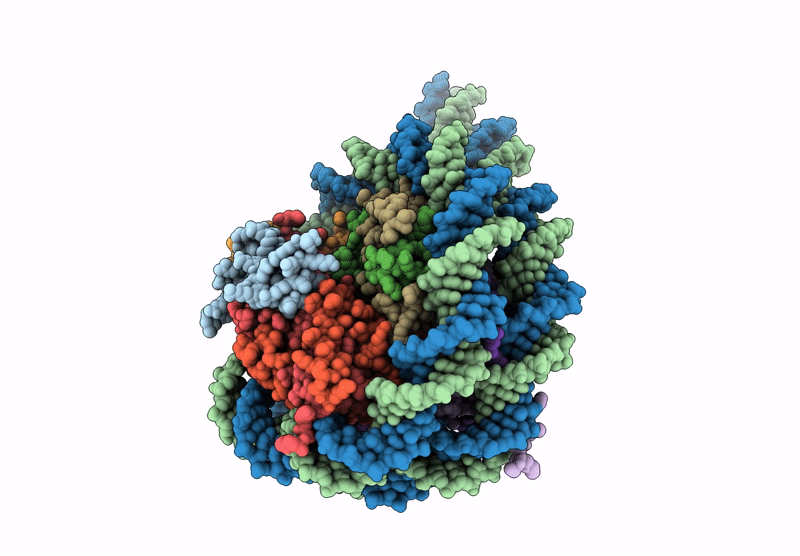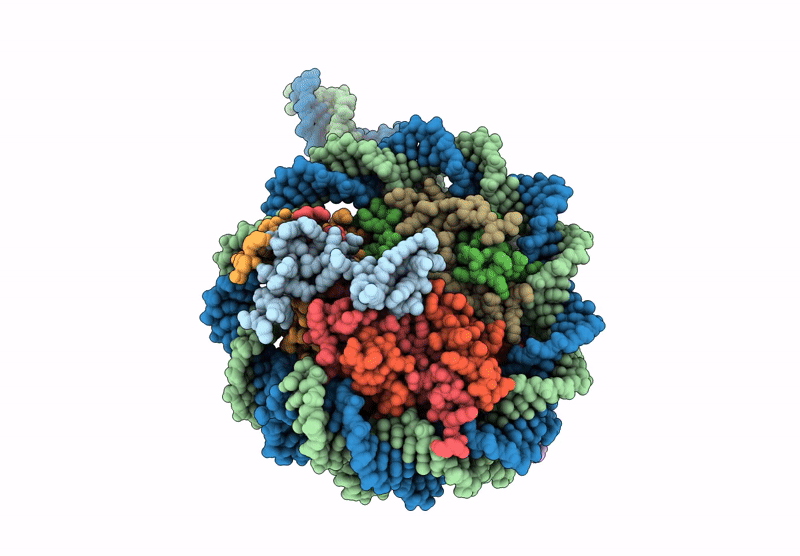
PDB ID:
9GFM
Keywords:
Title:
CryoEM structure of the human INO80 core-nucleosome complex state N-7
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
synthetic construct (Taxon ID: 32630)
synthetic construct (Taxon ID: 32630)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.80 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE