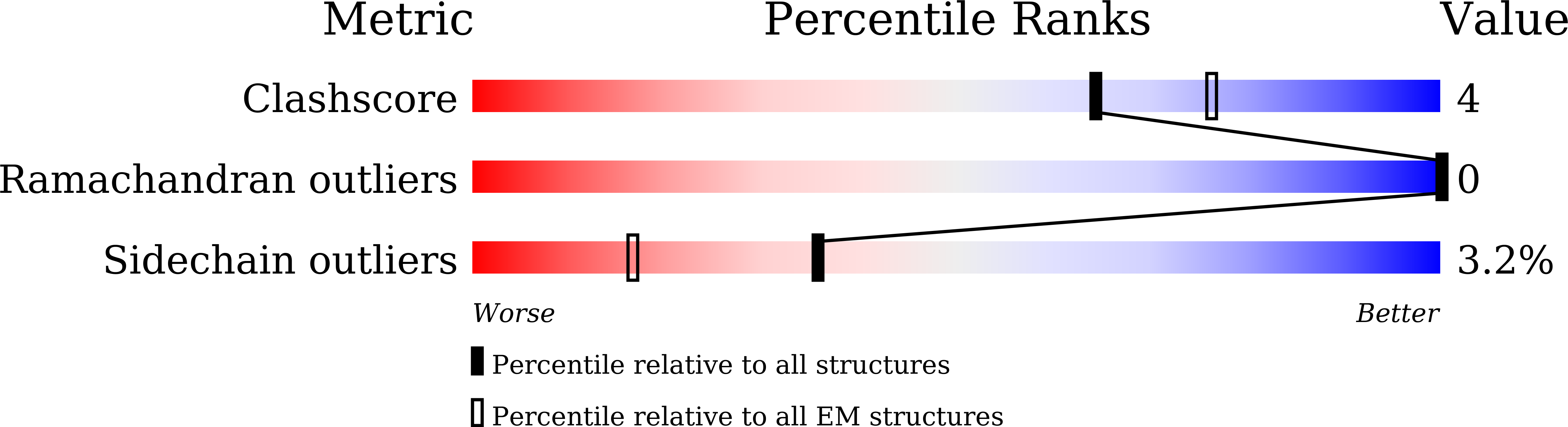
Deposition Date
2024-05-26
Release Date
2024-06-12
Last Version Date
2024-06-12
Entry Detail
PDB ID:
8ZNG
Keywords:
Title:
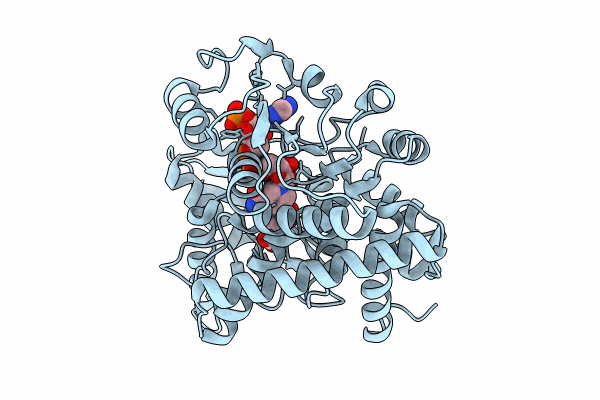
Cryo-EM structure of W89F mutated Glutamate dehydrogenase from Thermococcus profundus in complex with NADPH and AKG in the steady stage of reaction
Biological Source:
Source Organism(s):
Thermococcus profundus (Taxon ID: 49899)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.50 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE