Deposition Date
2024-03-01
Release Date
2025-03-12
Last Version Date
2026-03-25
Entry Detail
PDB ID:
8S7I
Keywords:
Title:
Fructose 6-phosphate aldolase, L107C/A129G/R134V/L163C/S166G mutant
Biological Source:
Source Organism(s):
Escherichia coli BL21 (Taxon ID: 511693)
Expression System(s):
Method Details:
Experimental Method:
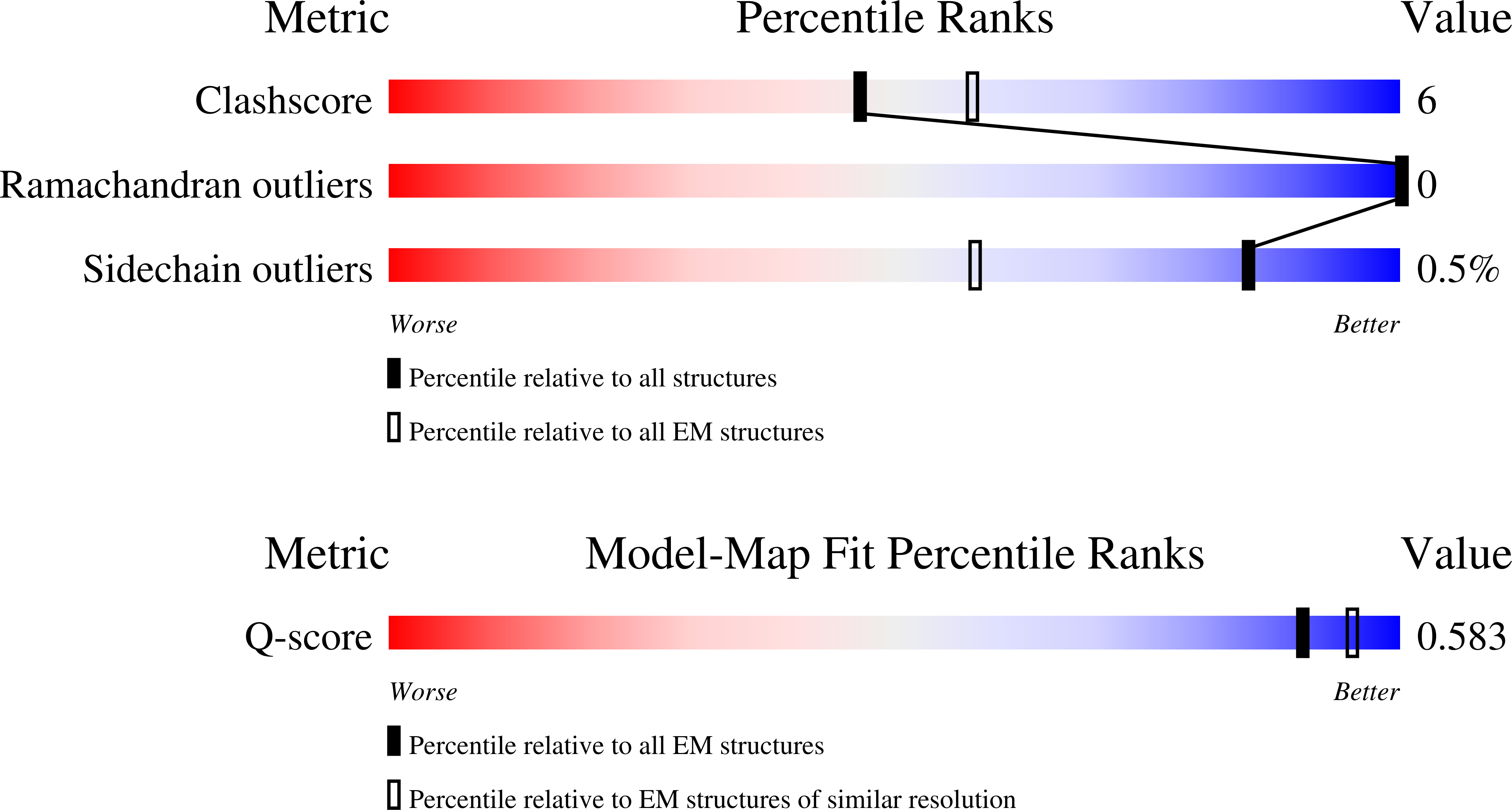
Resolution:
3.20 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE