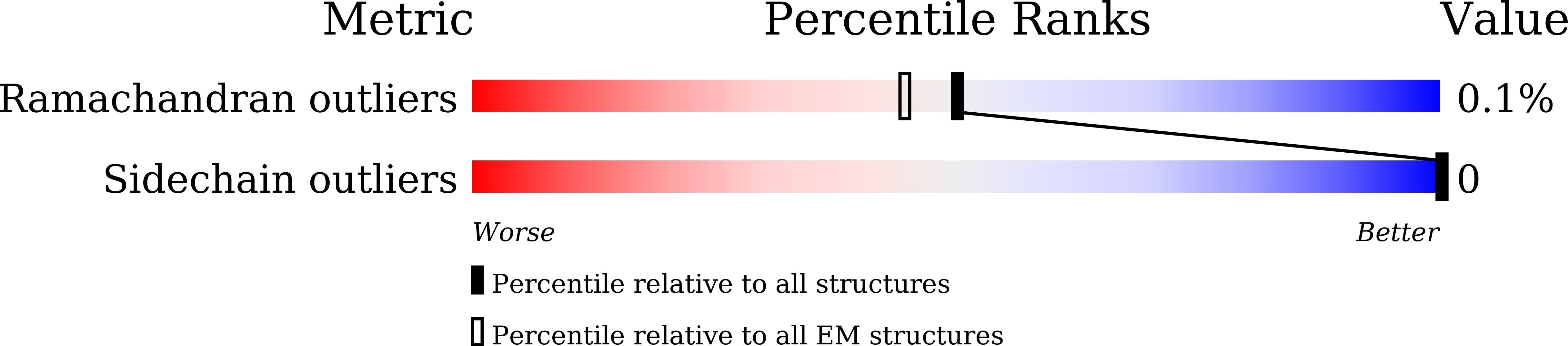
Deposition Date
2023-02-09
Release Date
2024-09-18
Last Version Date
2024-12-04
Entry Detail
PDB ID:
8IAQ
Keywords:
Title:
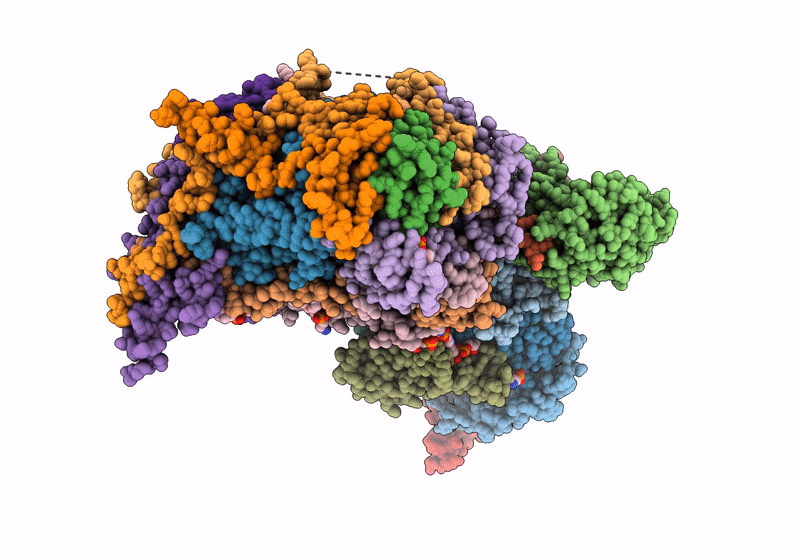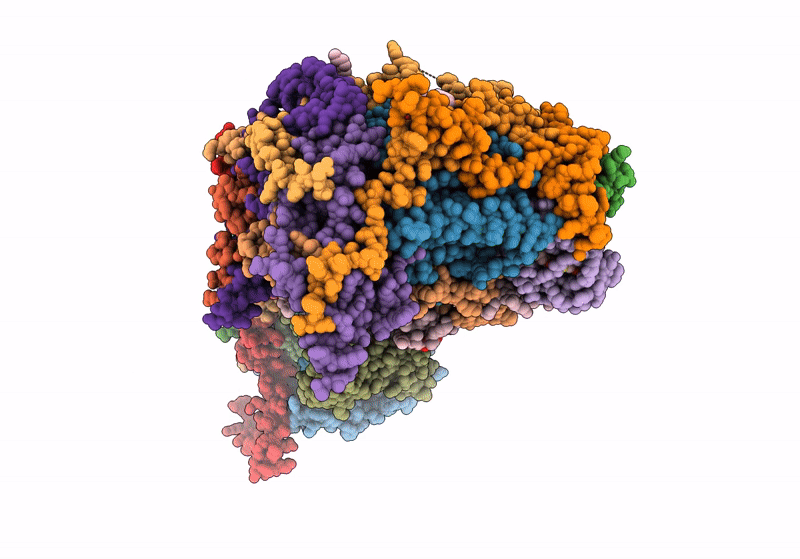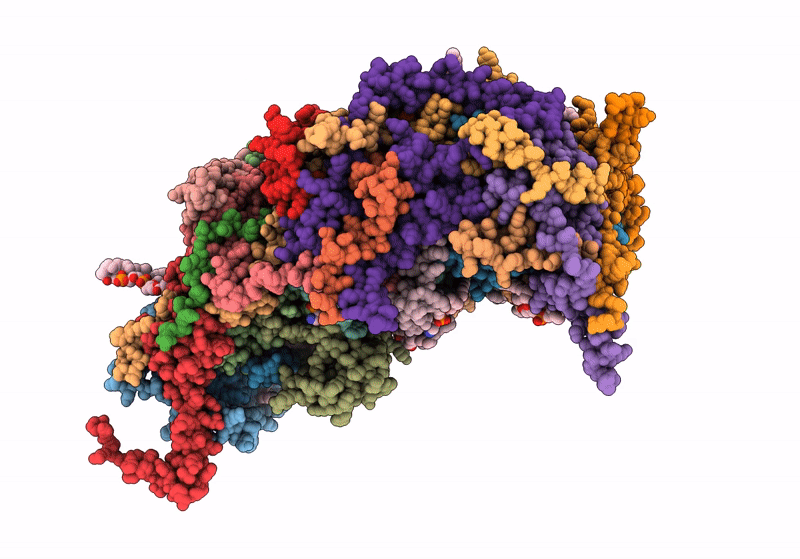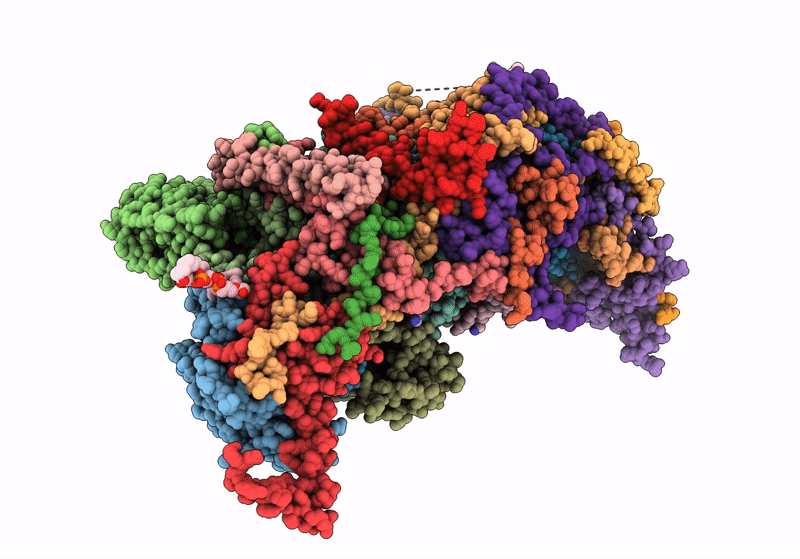
Respiratory complex Membrane domain of CI, focused map of type I, Wild type mouse under thermoneutral temperature
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Method Details:
Experimental Method:
Resolution:
3.40 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE