Deposition Date
2020-10-26
Release Date
2021-11-17
Last Version Date
2025-08-27
Entry Detail
PDB ID:
7KJN
Keywords:
Title:
CRYSTAL STRUCTURE OF HUMAN MDMX IN COMPLEX WITH D-PEPTIDE INHIBITOR (DPMI-OMEGA)
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
synthetic construct (Taxon ID: 32630)
synthetic construct (Taxon ID: 32630)
Method Details:
Experimental Method:
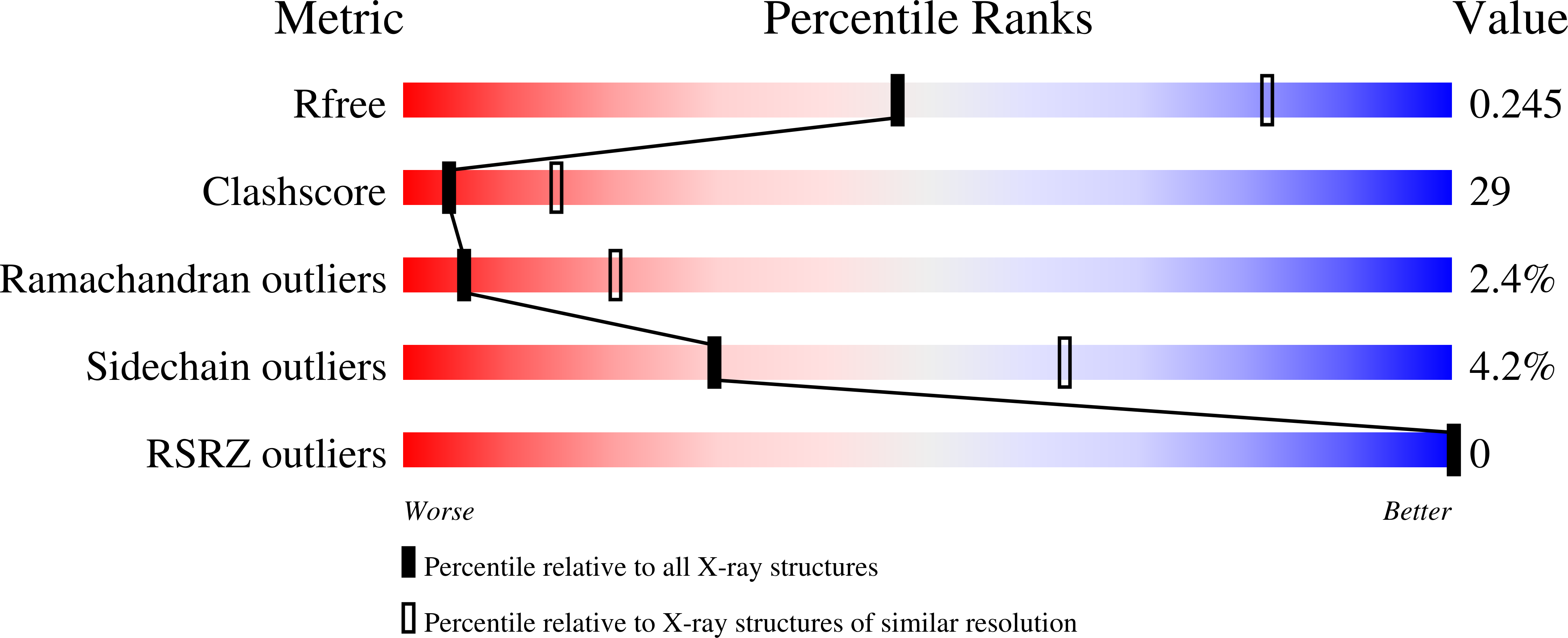
Resolution:
2.80 Å
R-Value Free:
0.24
R-Value Work:
0.20
R-Value Observed:
0.20
Space Group:
P 31 2 1