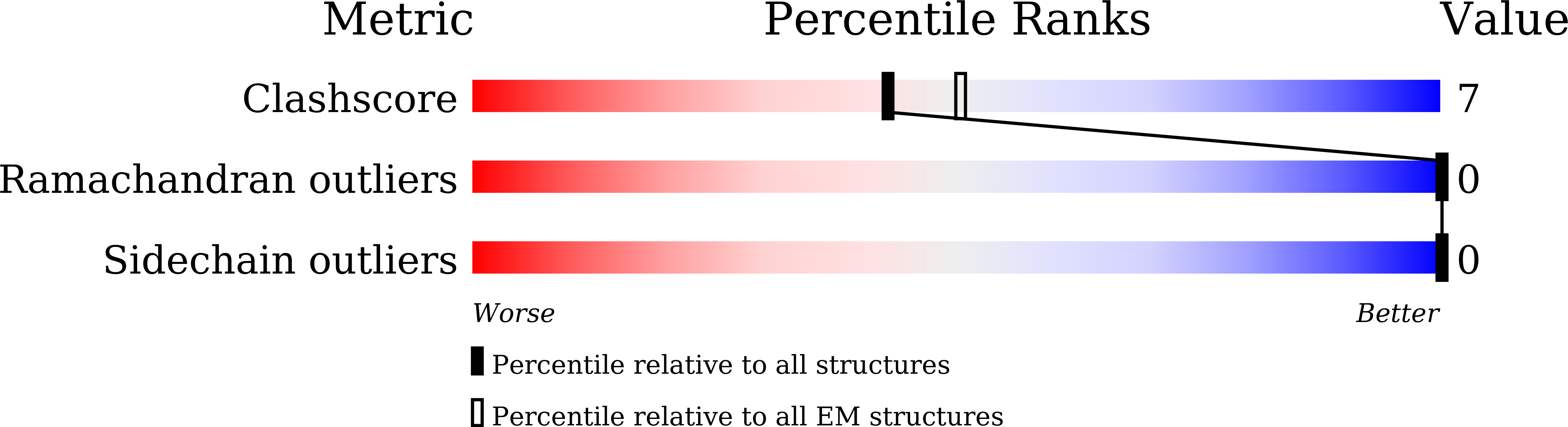
Deposition Date
2021-07-02
Release Date
2021-10-13
Last Version Date
2022-02-16
Entry Detail
PDB ID:
7F8V
Keywords:
Title:
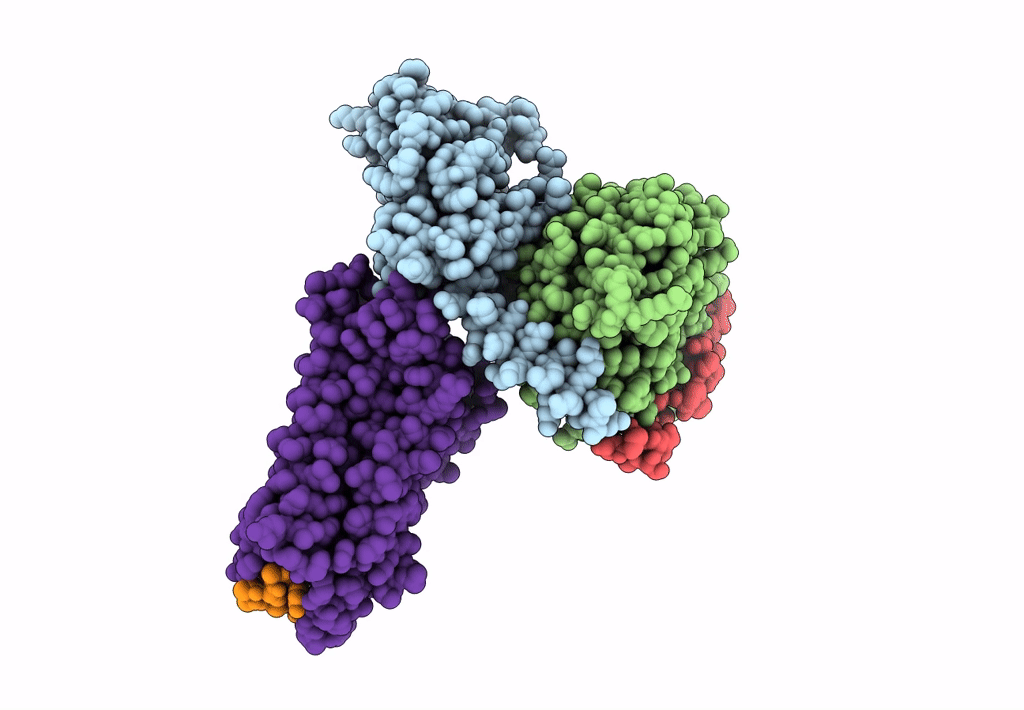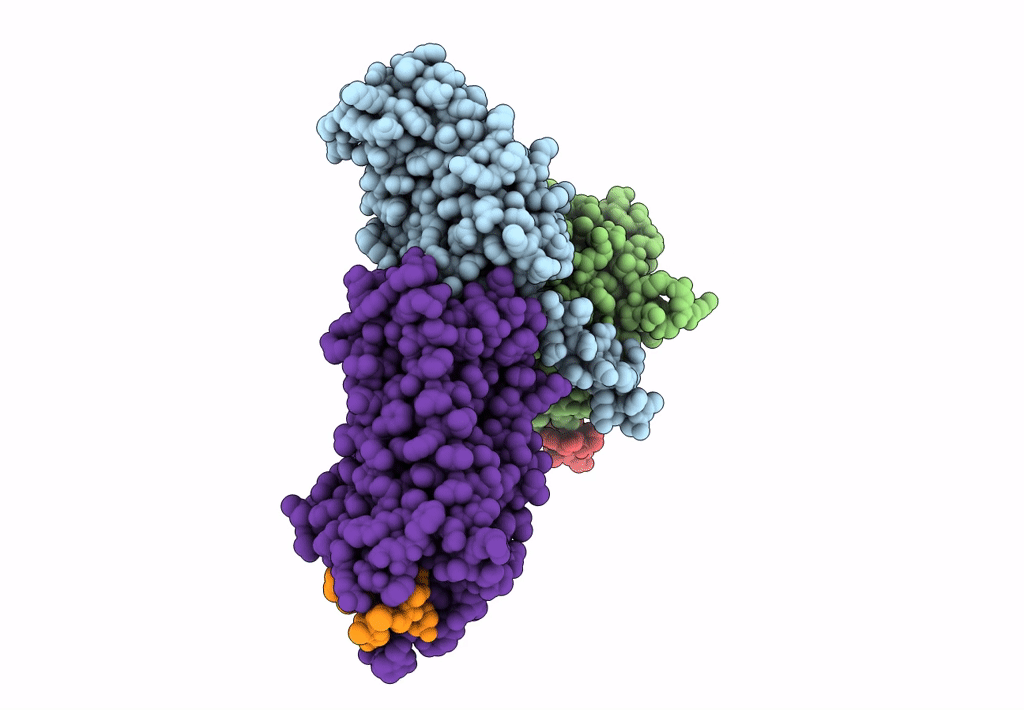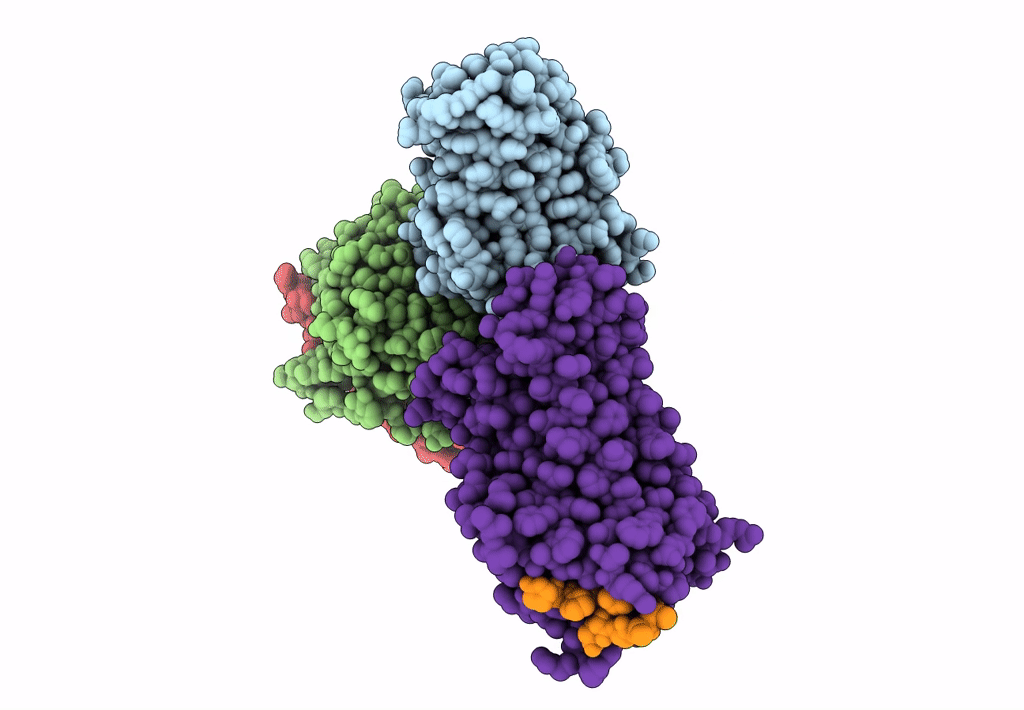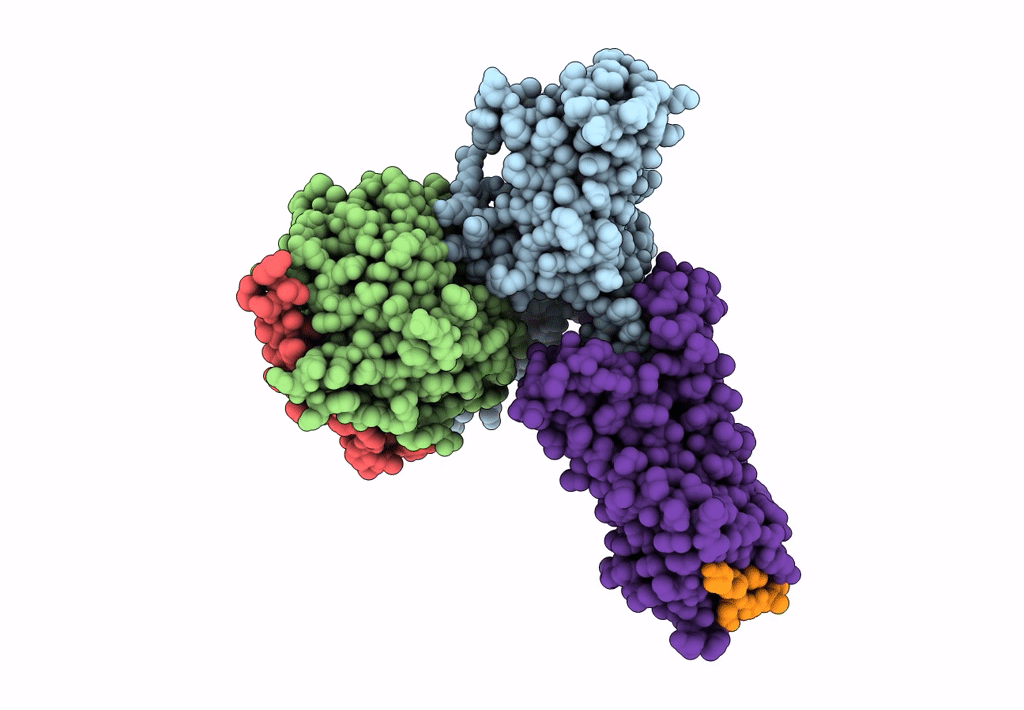
Cryo-EM structure of the cholecystokinin receptor CCKBR in complex with gastrin-17 and Gi
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.30 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE