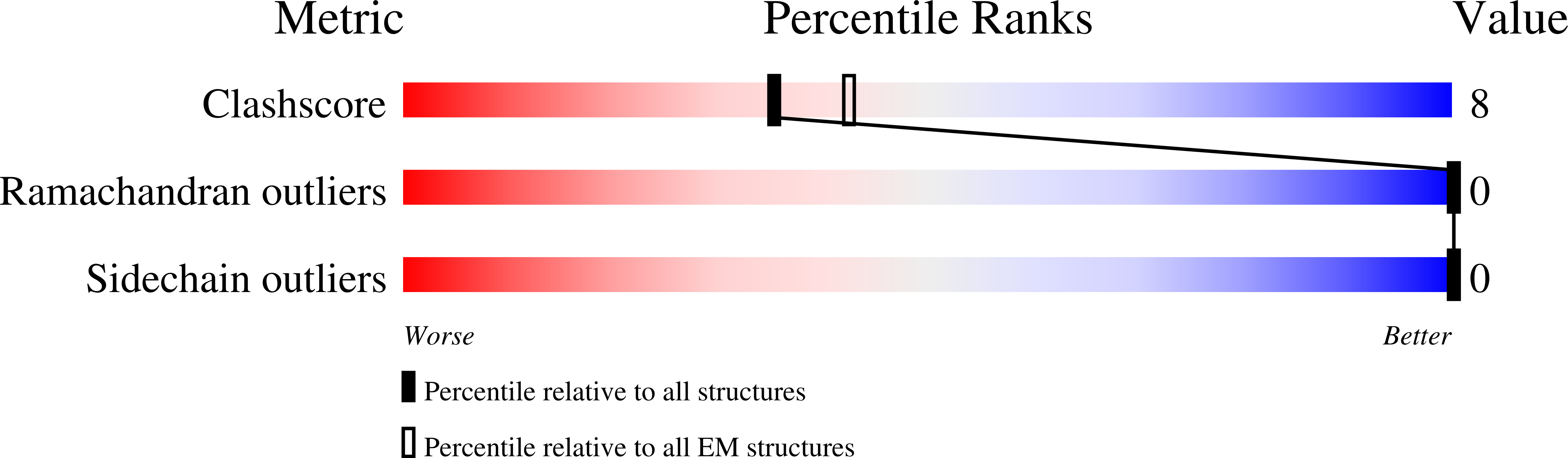
Deposition Date
2020-01-29
Release Date
2020-07-15
Last Version Date
2025-05-28
Entry Detail
PDB ID:
6VN1
Keywords:
Title:
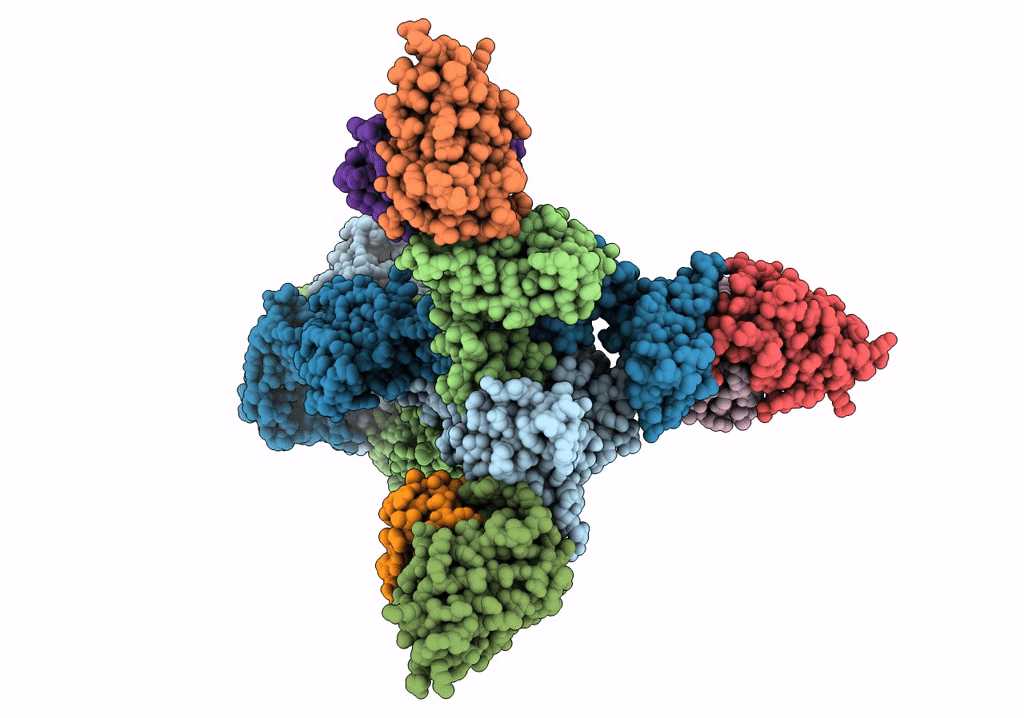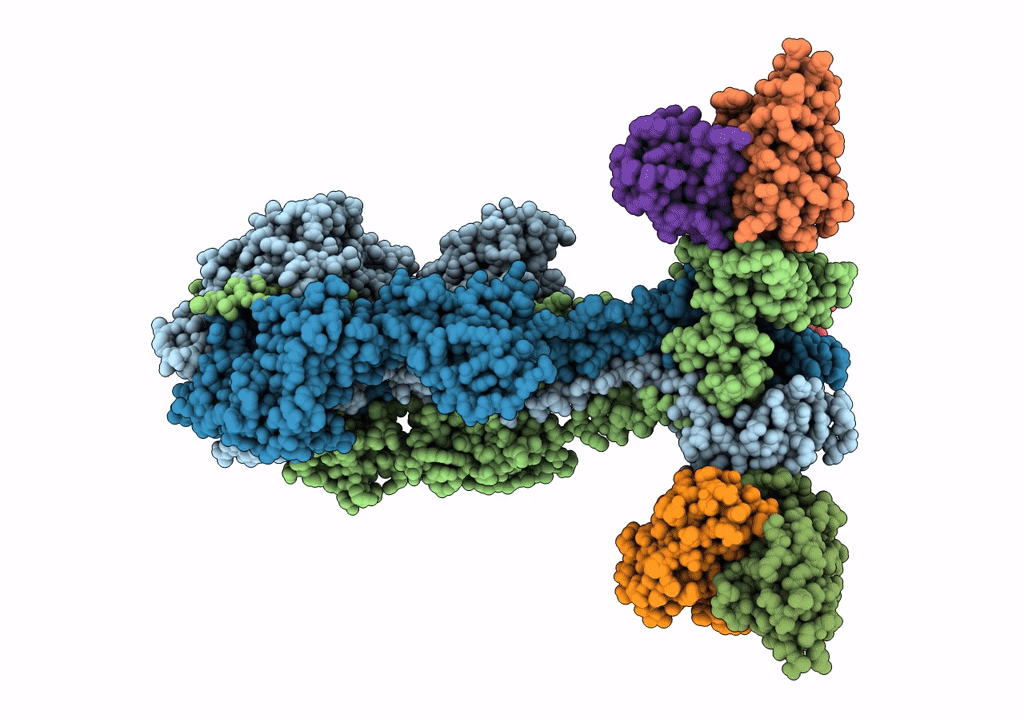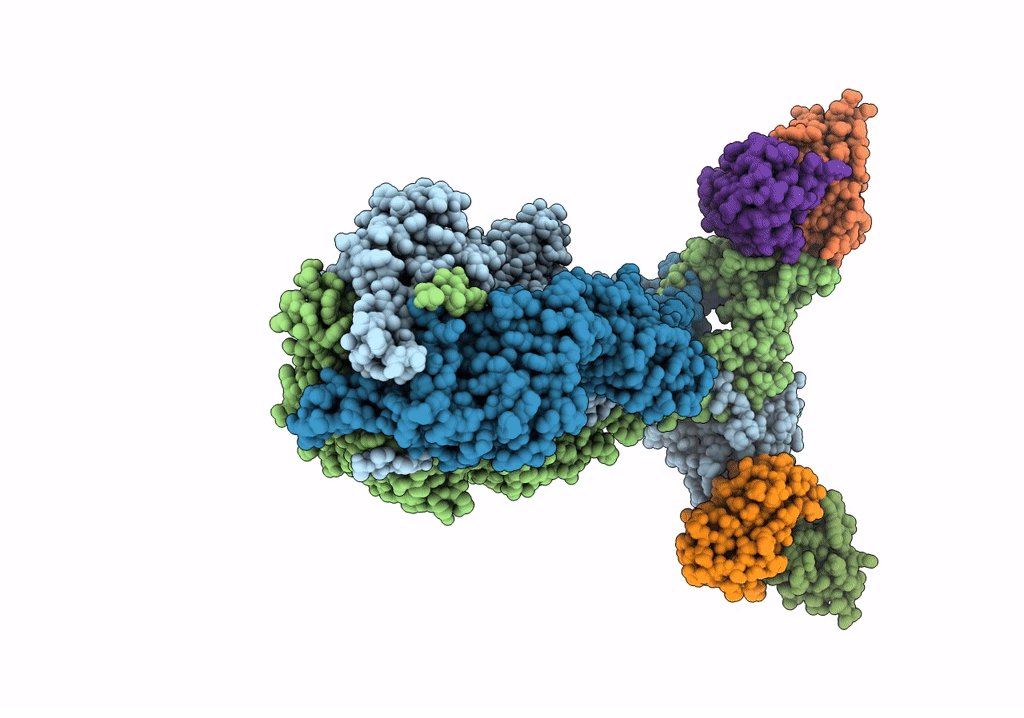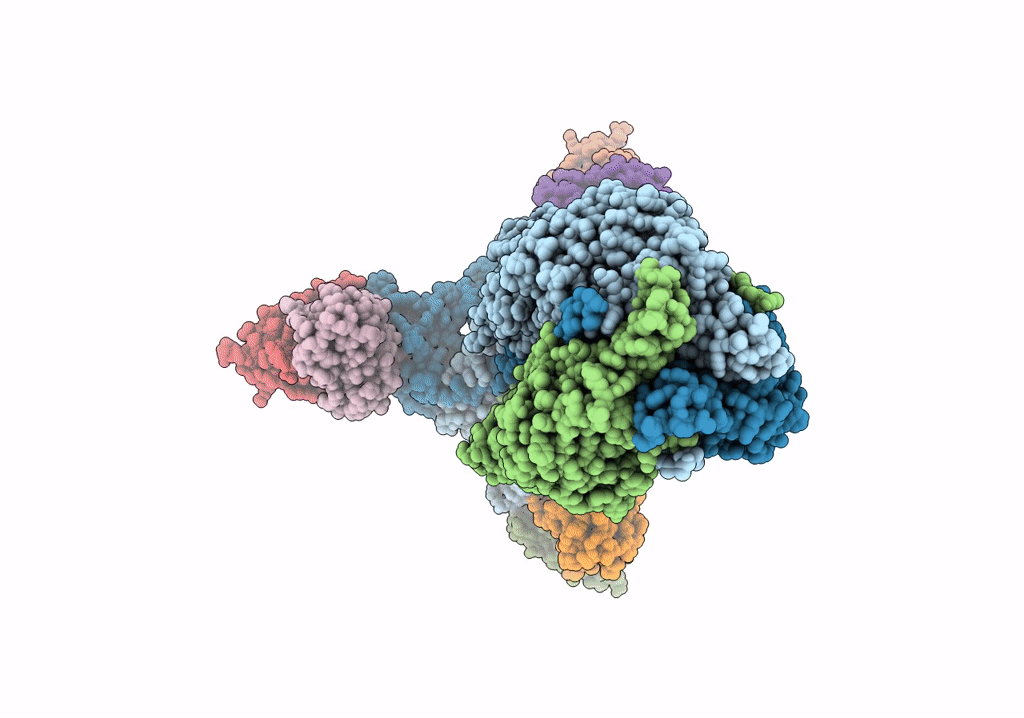
A 2.8 Angstrom Cryo-EM Structure of a Glycoprotein B-Neutralizing Antibody Complex Reveals a Critical Domain for Herpesvirus Fusion Initiation
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Human alphaherpesvirus 3 (Taxon ID: 10335)
Human alphaherpesvirus 3 (Taxon ID: 10335)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.80 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE