Deposition Date
2019-10-18
Release Date
2020-06-10
Last Version Date
2023-10-11
Entry Detail
PDB ID:
6UPY
Keywords:
Title:
RNA polymerase II elongation complex with 5-guanidinohydantoin lesion in state 2E
Biological Source:
Source Organism(s):
synthetic construct (Taxon ID: 32630)
Saccharomyces cerevisiae (strain ATCC 204508 / S288c) (Taxon ID: 559292)
Saccharomyces cerevisiae (strain ATCC 204508 / S288c) (Taxon ID: 559292)
Method Details:
Experimental Method:
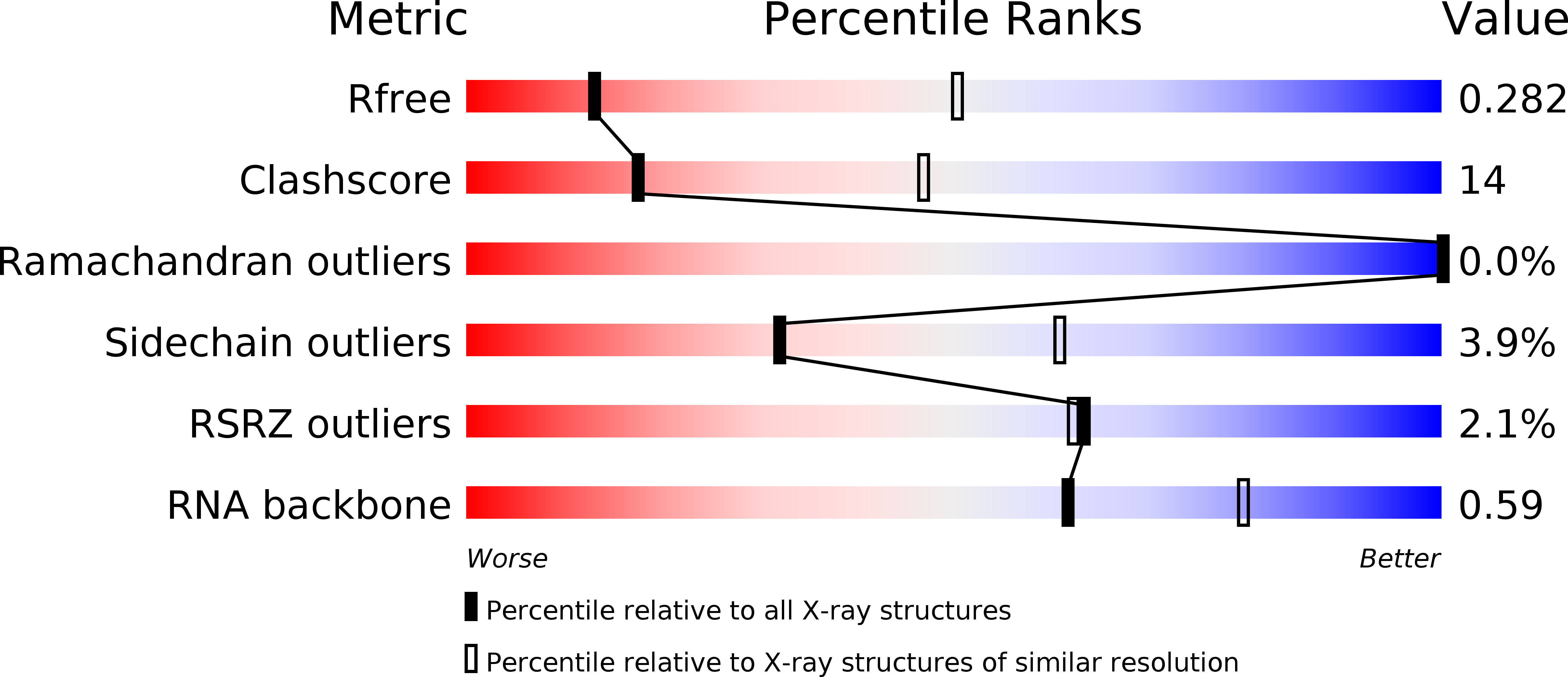
Resolution:
3.40 Å
R-Value Free:
0.28
R-Value Work:
0.22
R-Value Observed:
0.22
Space Group:
C 1 2 1