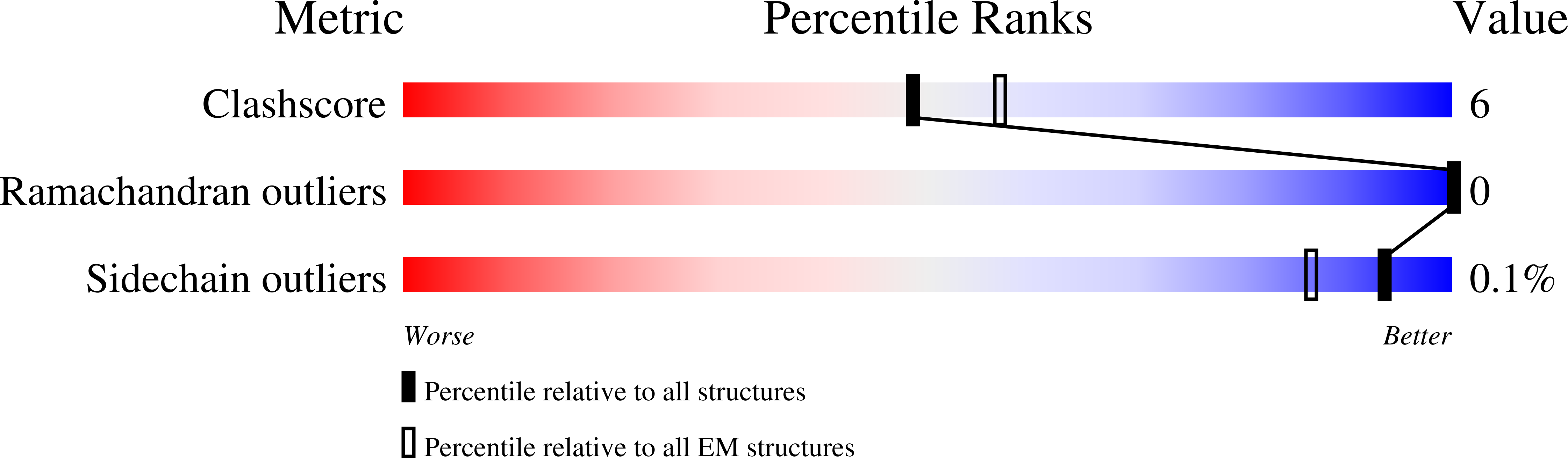Deposition Date
2019-09-24
Release Date
2019-11-20
Last Version Date
2024-07-10
Entry Detail
PDB ID:
6SWY
Keywords:
Title:
Structure of active GID E3 ubiquitin ligase complex minus Gid2 and delta Gid9 RING domain
Biological Source:
Source Organism(s):
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.20 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE