Deposition Date
2018-05-21
Release Date
2018-07-25
Last Version Date
2024-10-16
Entry Detail
PDB ID:
5ZXV
Keywords:
Title:
Structural definition of a unique neutralization epitope on the receptor-binding domain of MERS-CoV spike glycoprotein
Biological Source:
Source Organism(s):
Middle East respiratory syndrome coronavirus (Taxon ID: 1335626)
Homo sapiens (Taxon ID: 9606)
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
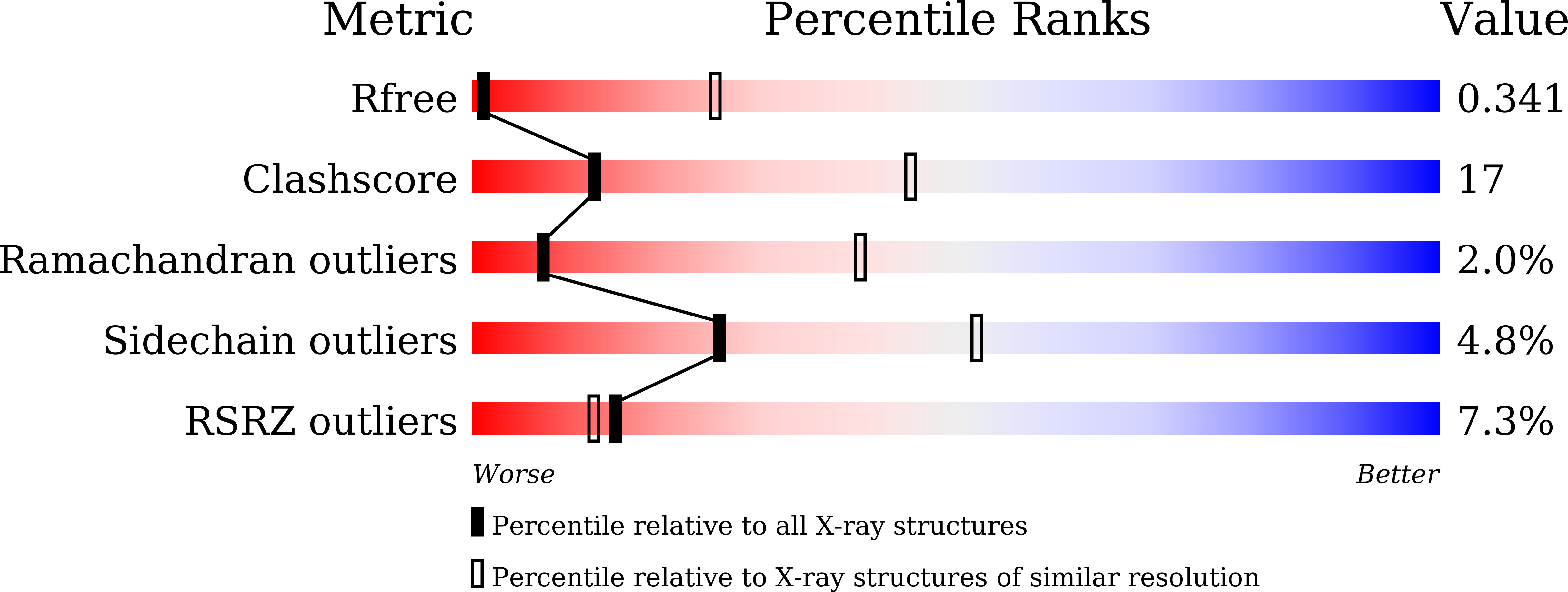
Resolution:
4.48 Å
R-Value Free:
0.34
R-Value Work:
0.30
R-Value Observed:
0.31
Space Group:
H 3