Deposition Date
2012-09-04
Release Date
2013-01-16
Last Version Date
2023-09-13
Entry Detail
PDB ID:
4GXK
Keywords:
Title:
R283K DNA polymerase beta ternary complex with a templating 8OG and incoming dATP analog
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
synthetic construct (Taxon ID: 32630)
synthetic construct (Taxon ID: 32630)
Expression System(s):
Method Details:
Experimental Method:
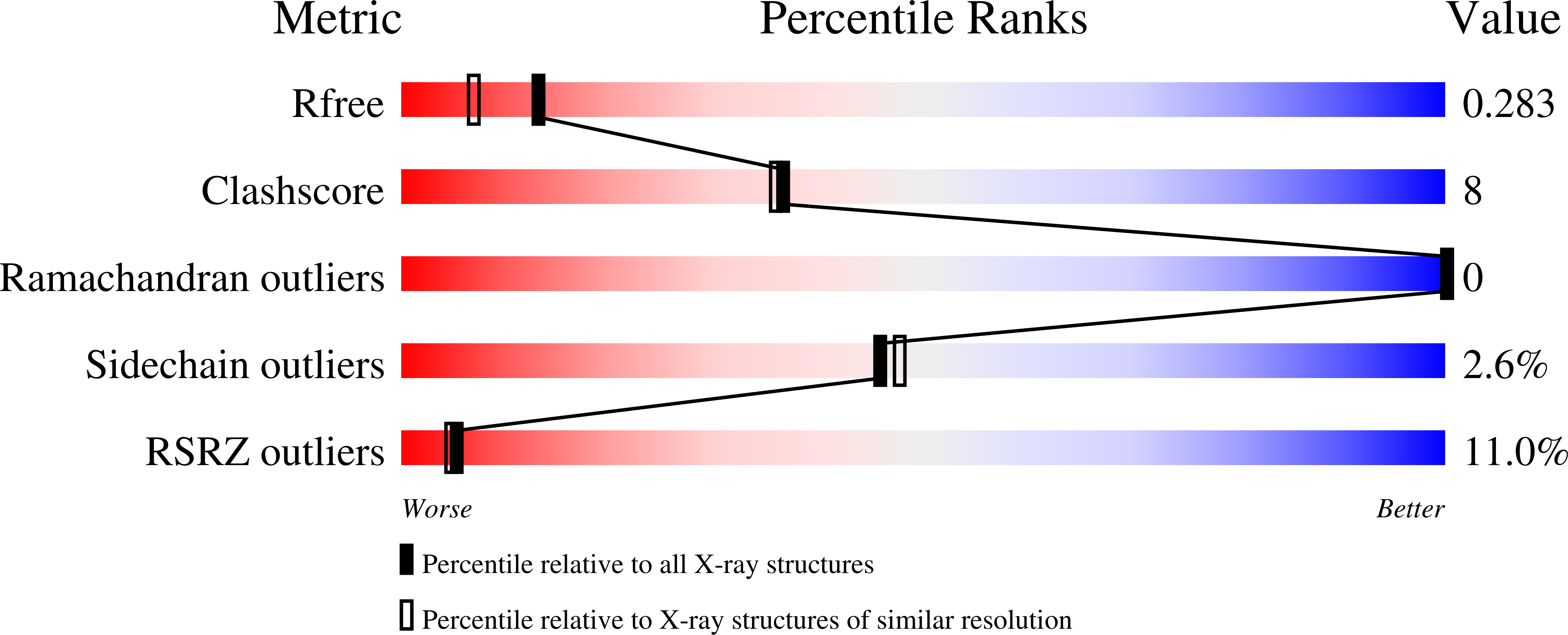
Resolution:
2.00 Å
R-Value Free:
0.27
R-Value Work:
0.21
R-Value Observed:
0.22
Space Group:
P 1 21 1