Deposition Date
2012-04-05
Release Date
2012-05-23
Last Version Date
2023-09-13
Entry Detail
PDB ID:
4EIR
Keywords:
Title:
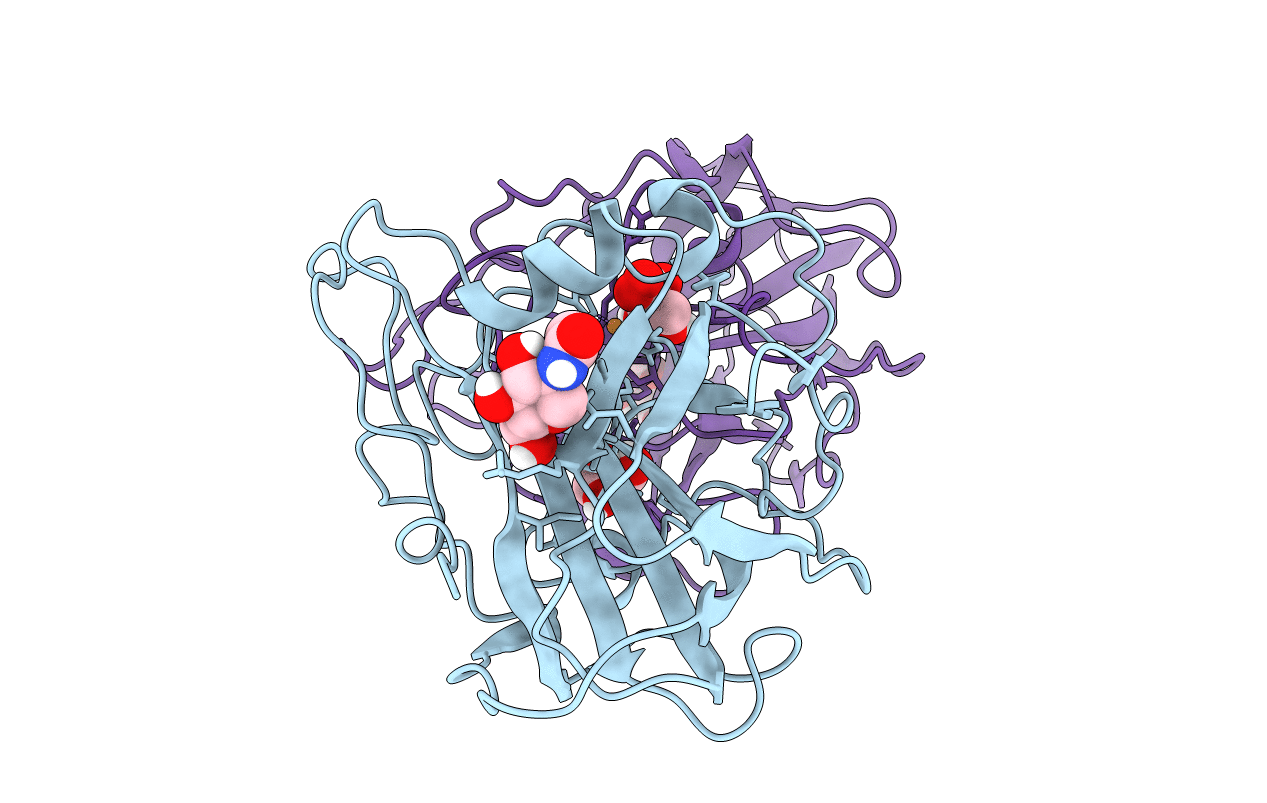
Structural basis for substrate targeting and catalysis by fungal polysaccharide monooxygenases (PMO-2)
Biological Source:
Source Organism(s):
Neurospora crassa (Taxon ID: 367110)
Method Details:
Experimental Method:
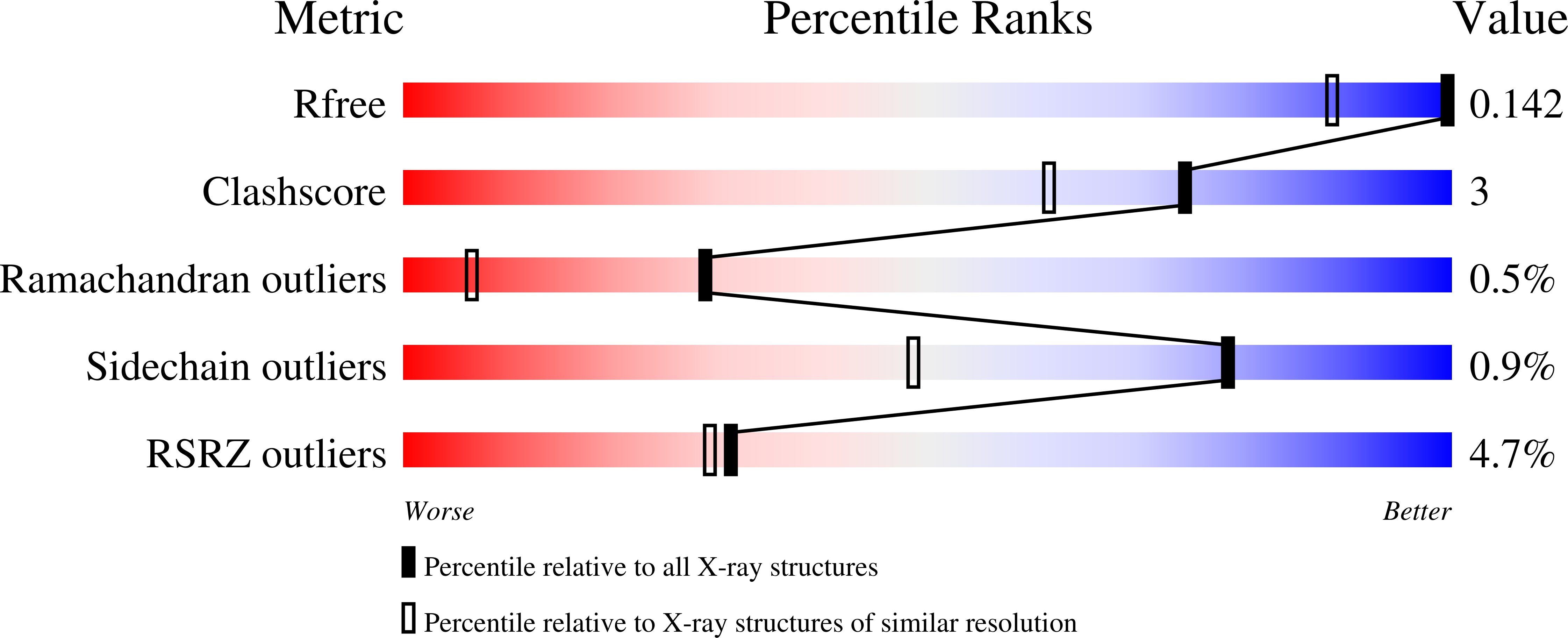
Resolution:
1.10 Å
R-Value Free:
0.14
R-Value Work:
0.13
R-Value Observed:
0.13
Space Group:
P 1 21 1