Deposition Date
2016-04-08
Release Date
2016-07-13
Last Version Date
2025-06-11
Entry Detail
PDB ID:
3JD5
Keywords:
Title:
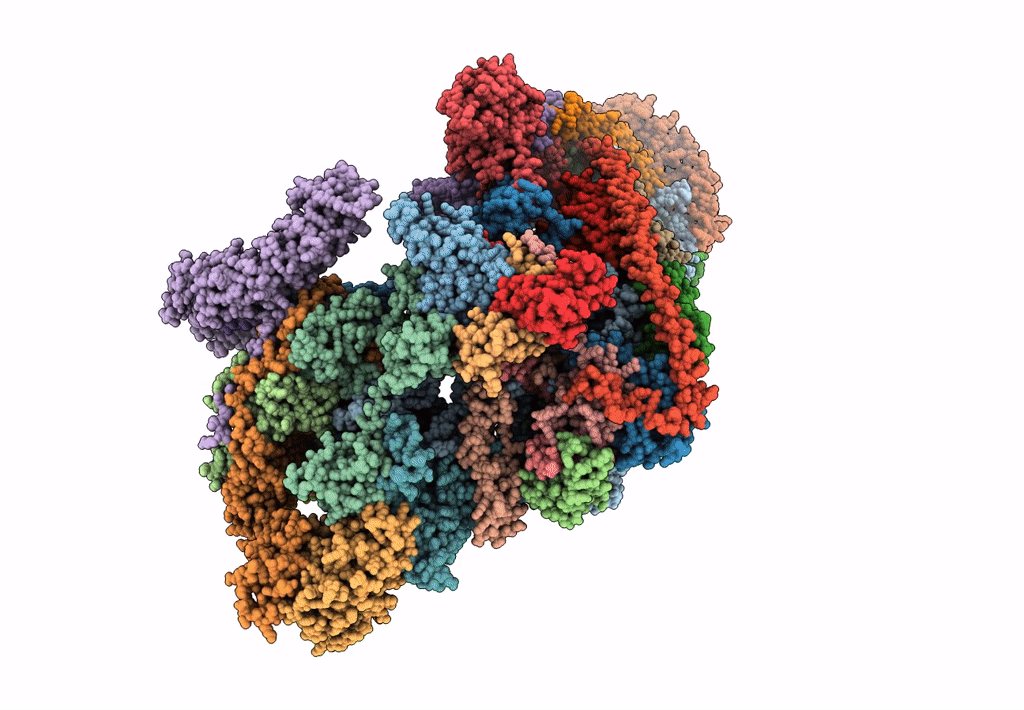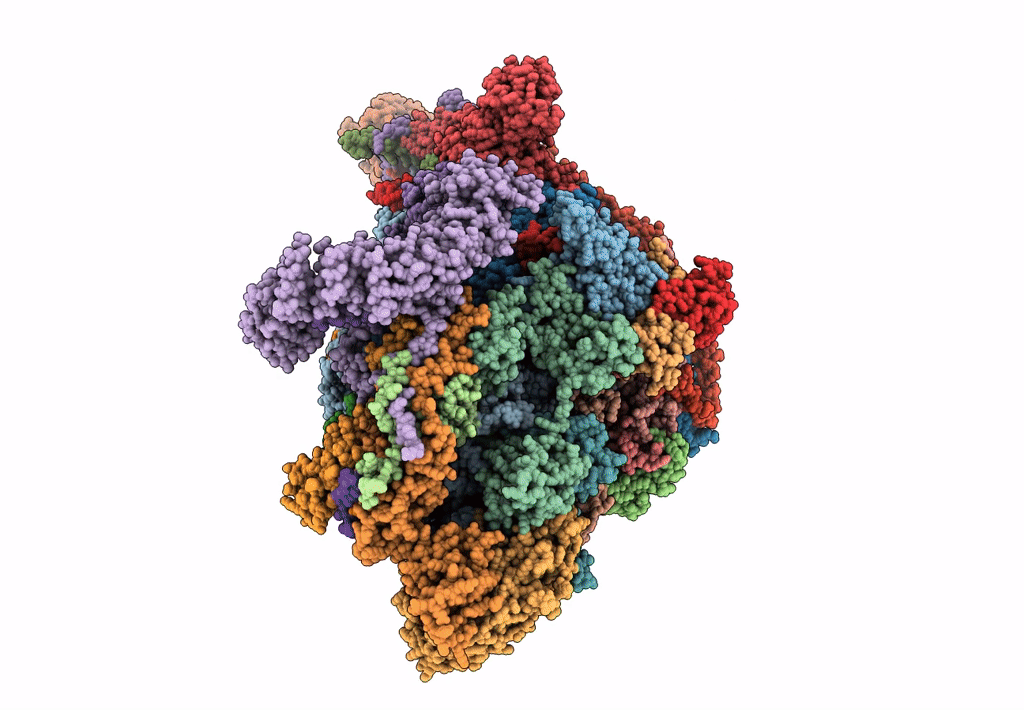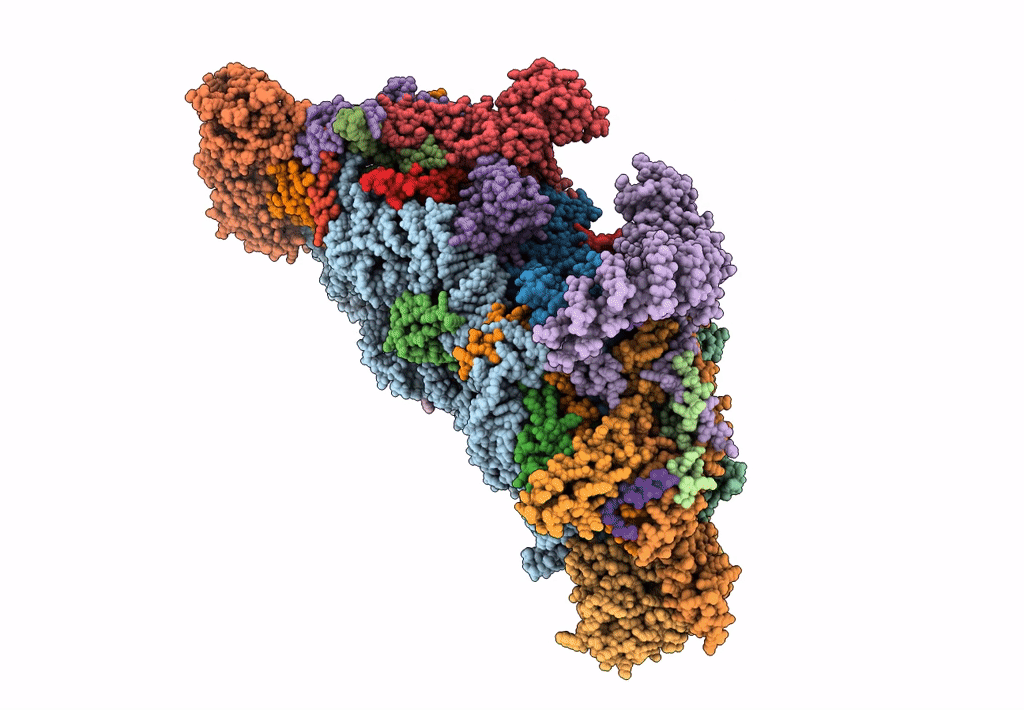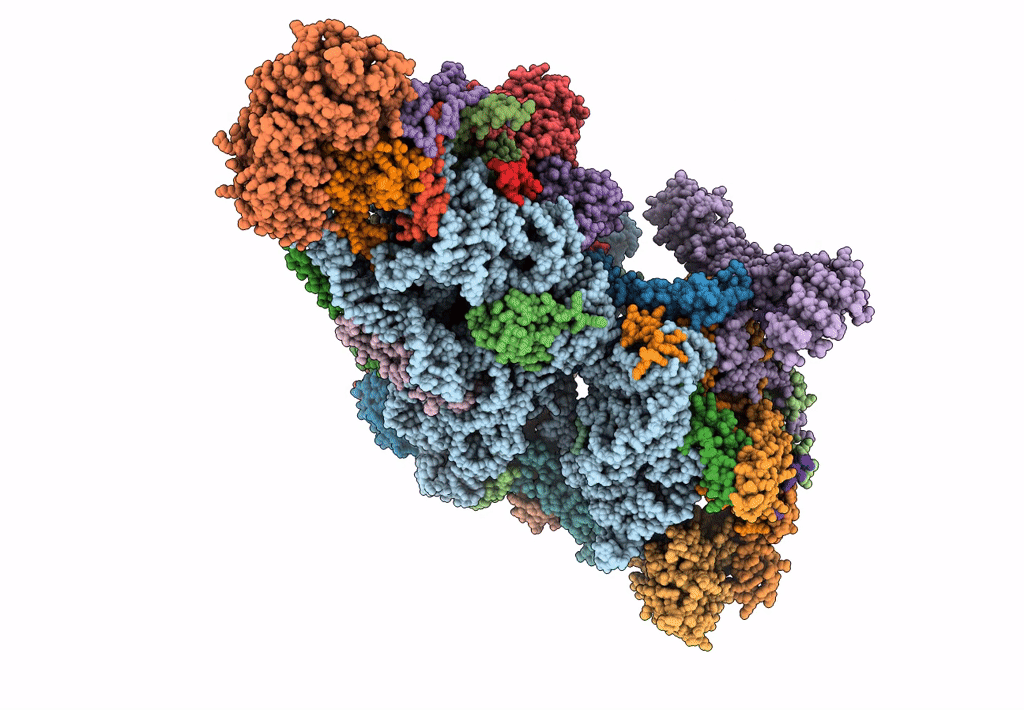
Cryo-EM structure of the small subunit of the mammalian mitochondrial ribosome
Biological Source:
Source Organism(s):
Bos taurus (Taxon ID: 9913)
Method Details:
Experimental Method:
Resolution:
7.00 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE