Deposition Date
2008-11-19
Release Date
2009-10-06
Last Version Date
2023-11-01
Entry Detail
PDB ID:
2ZVT
Keywords:
Title:
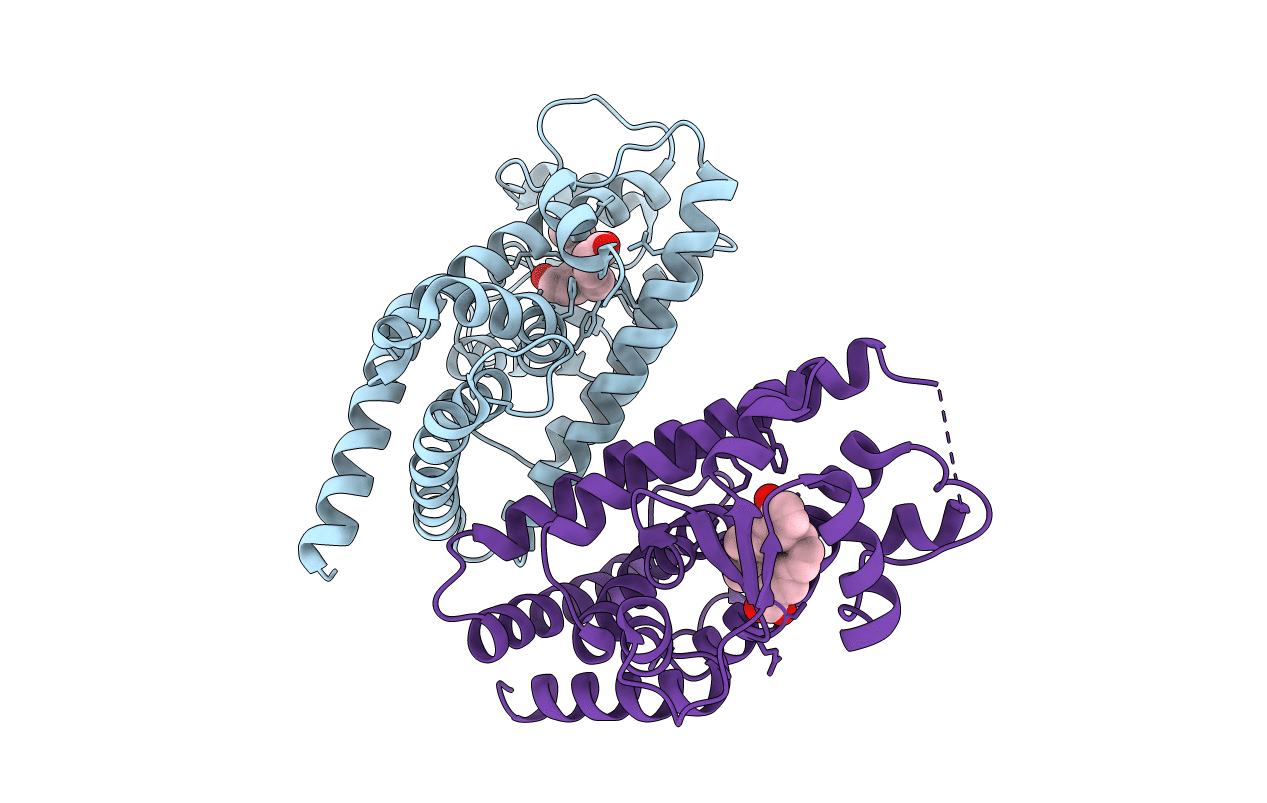
Cys285Ser mutant PPARgamma ligand-binding domain complexed with 15-deoxy-delta12,14-prostaglandin J2
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
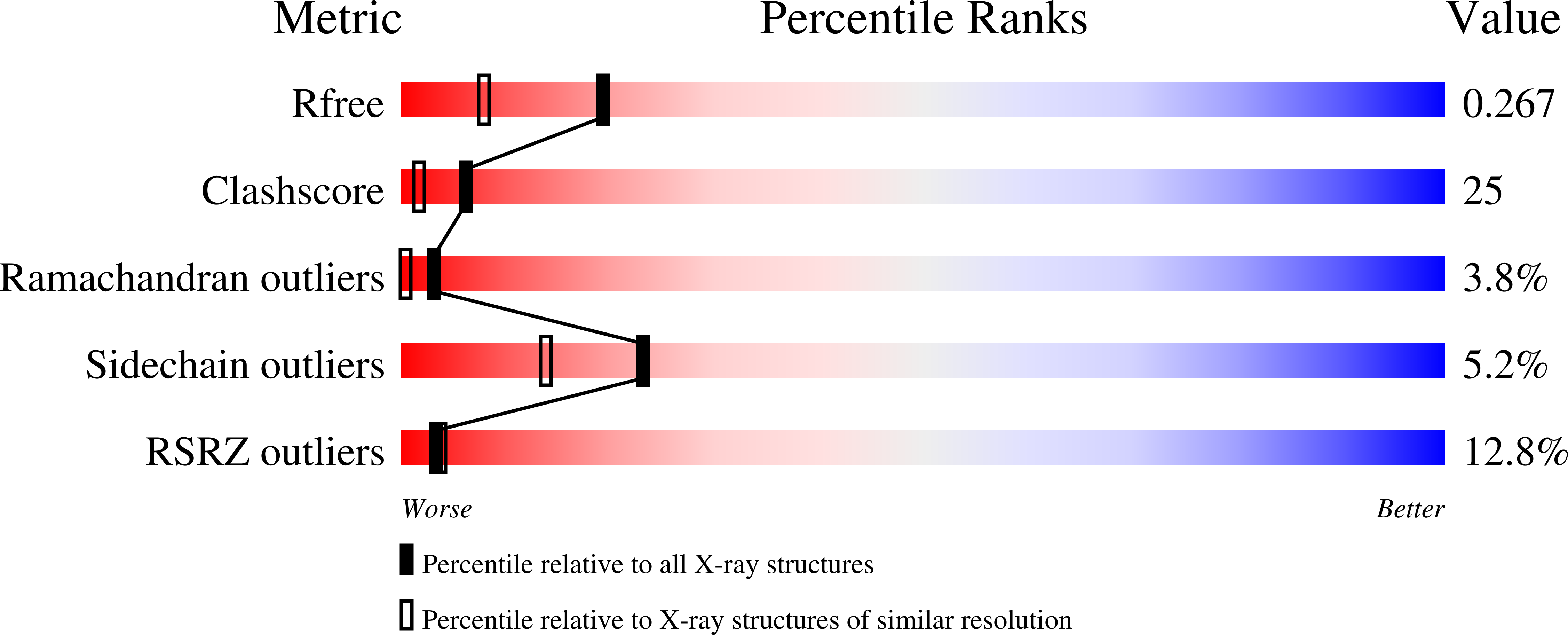
Resolution:
1.90 Å
R-Value Free:
0.27
R-Value Work:
0.25
R-Value Observed:
0.25
Space Group:
C 1 2 1