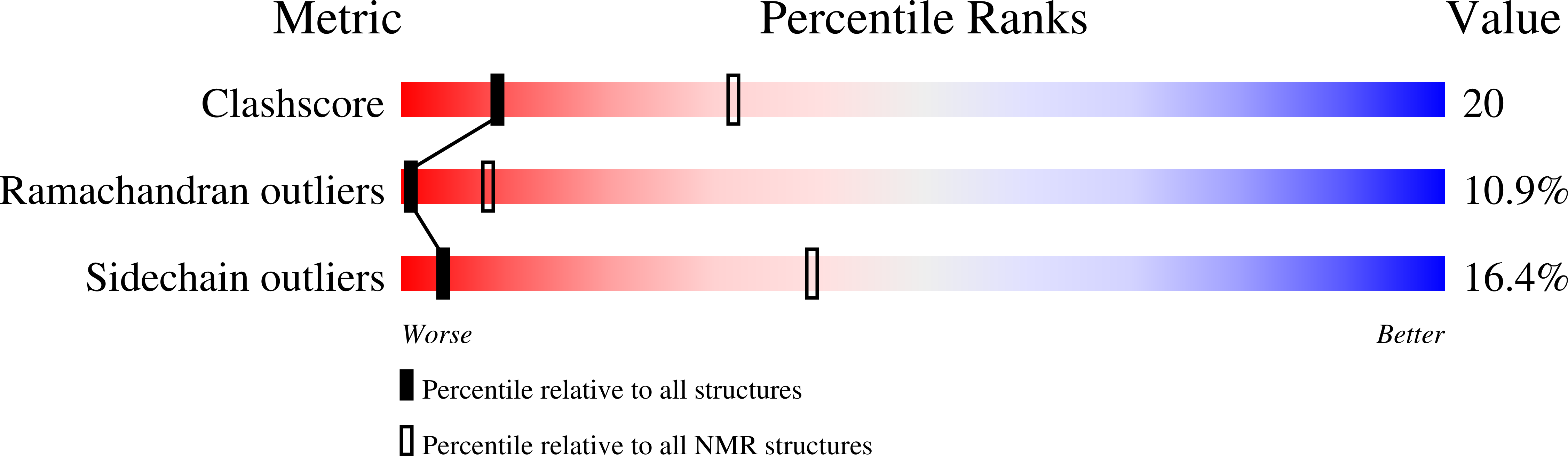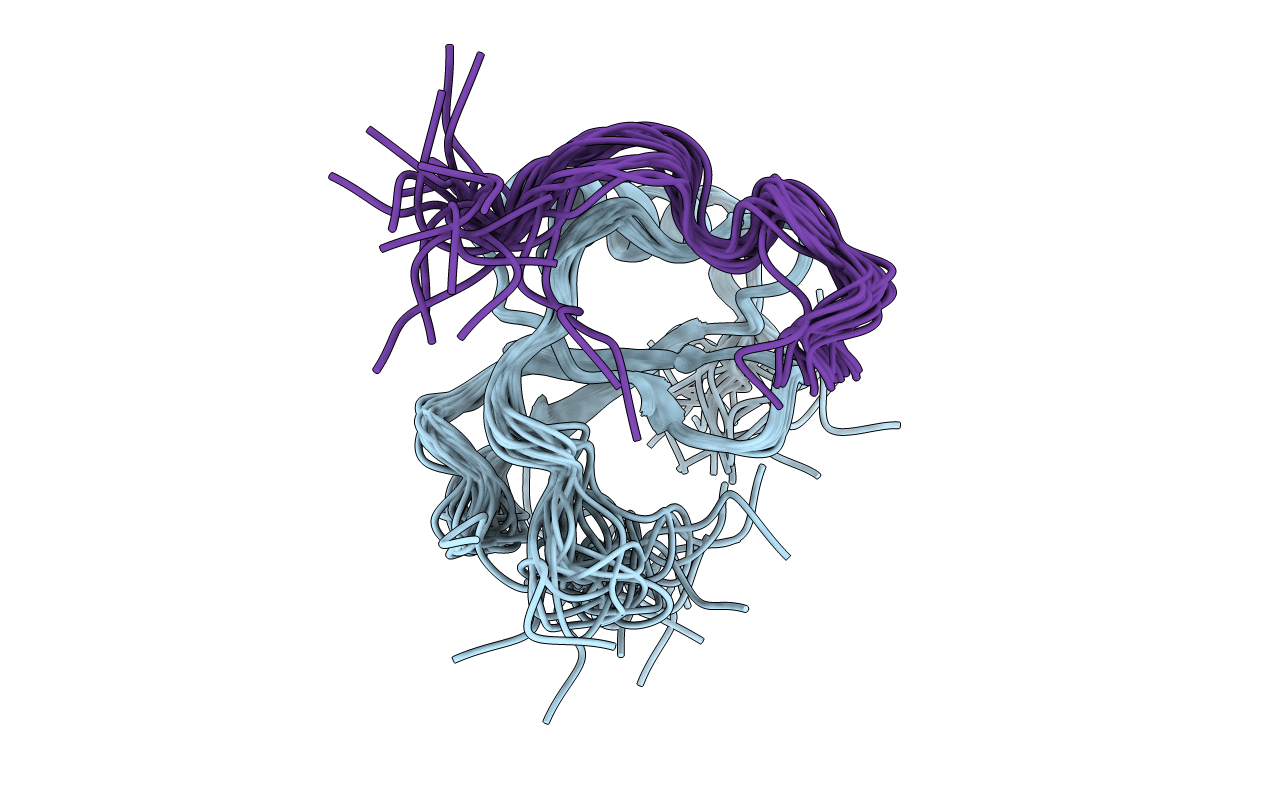
Deposition Date
2014-05-26
Release Date
2014-12-10
Last Version Date
2024-11-06
Entry Detail
PDB ID:
2MPM
Keywords:
Title:
Structural Basis of Receptor Sulfotyrosine Recognition by a CC Chemokine: the N-terminal Region of CCR3 Bound to CCL11/Eotaxin-1
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
64
Conformers Submitted:
20
Selection Criteria:
structures with the lowest energy