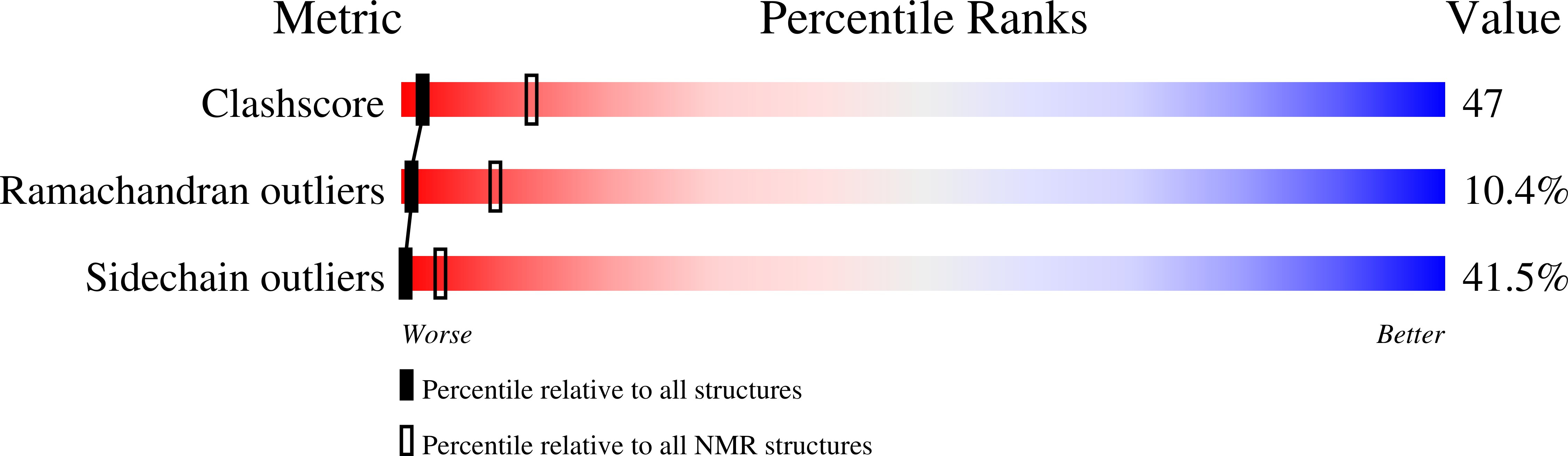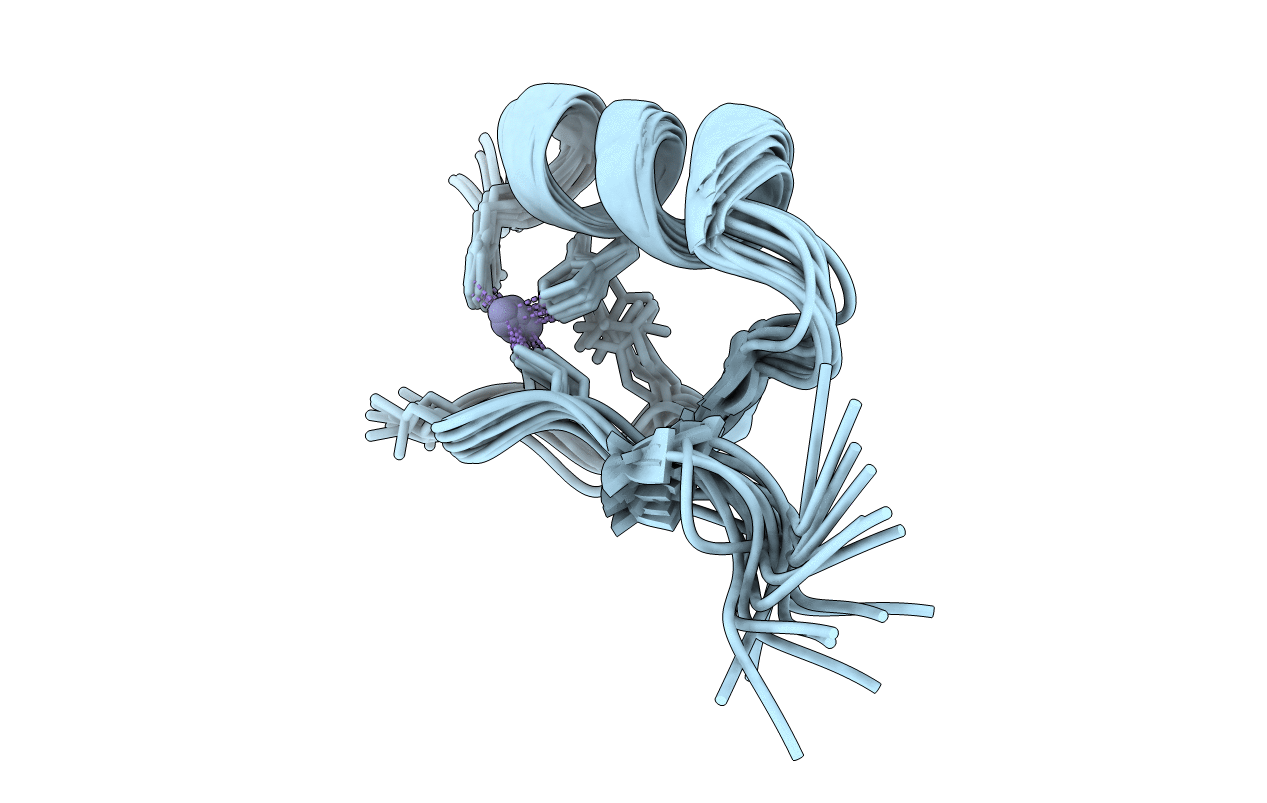
Deposition Date
2010-03-15
Release Date
2010-04-14
Last Version Date
2024-11-20
Entry Detail
PDB ID:
2KVH
Keywords:
Title:
Structure of the three-Cys2His2 domain of mouse testis zinc finger protein
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
50
Conformers Submitted:
15
Selection Criteria:
structures with the lowest energy