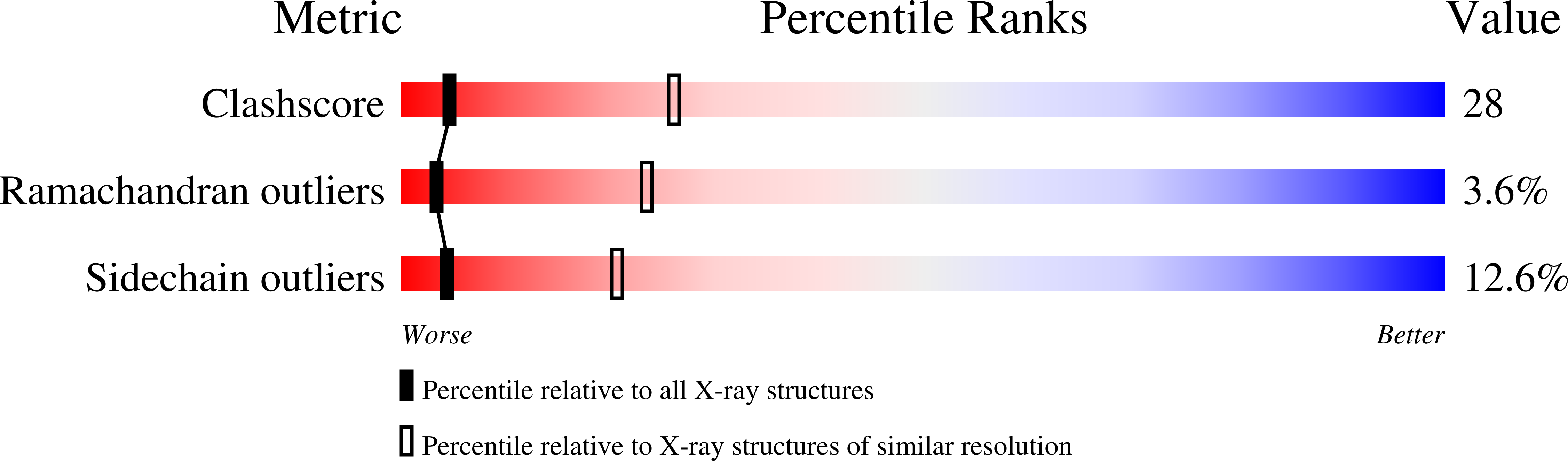
Deposition Date
2006-03-02
Release Date
2007-01-02
Last Version Date
2023-08-30
Entry Detail
PDB ID:
2G8G
Keywords:
Title:
Structurally mapping the diverse phenotype of Adeno-Associated Virus serotype 4
Biological Source:
Source Organism(s):
Adeno-associated virus - 4 (Taxon ID: 57579)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.20 Å
R-Value Free:
0.27
R-Value Work:
0.26
Space Group:
I 2 2 2