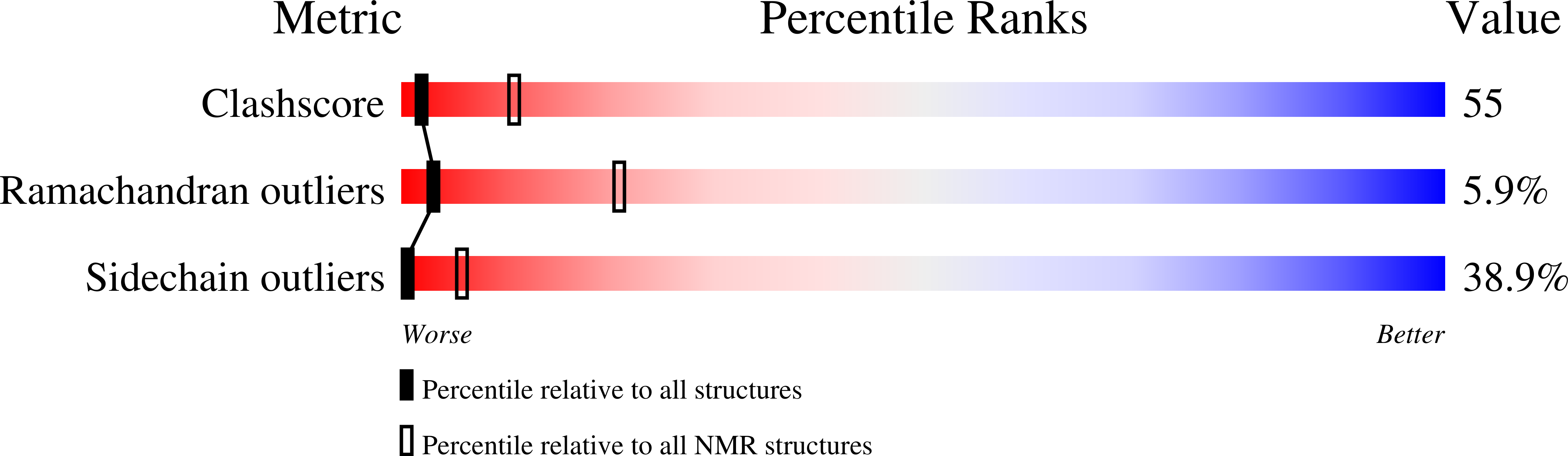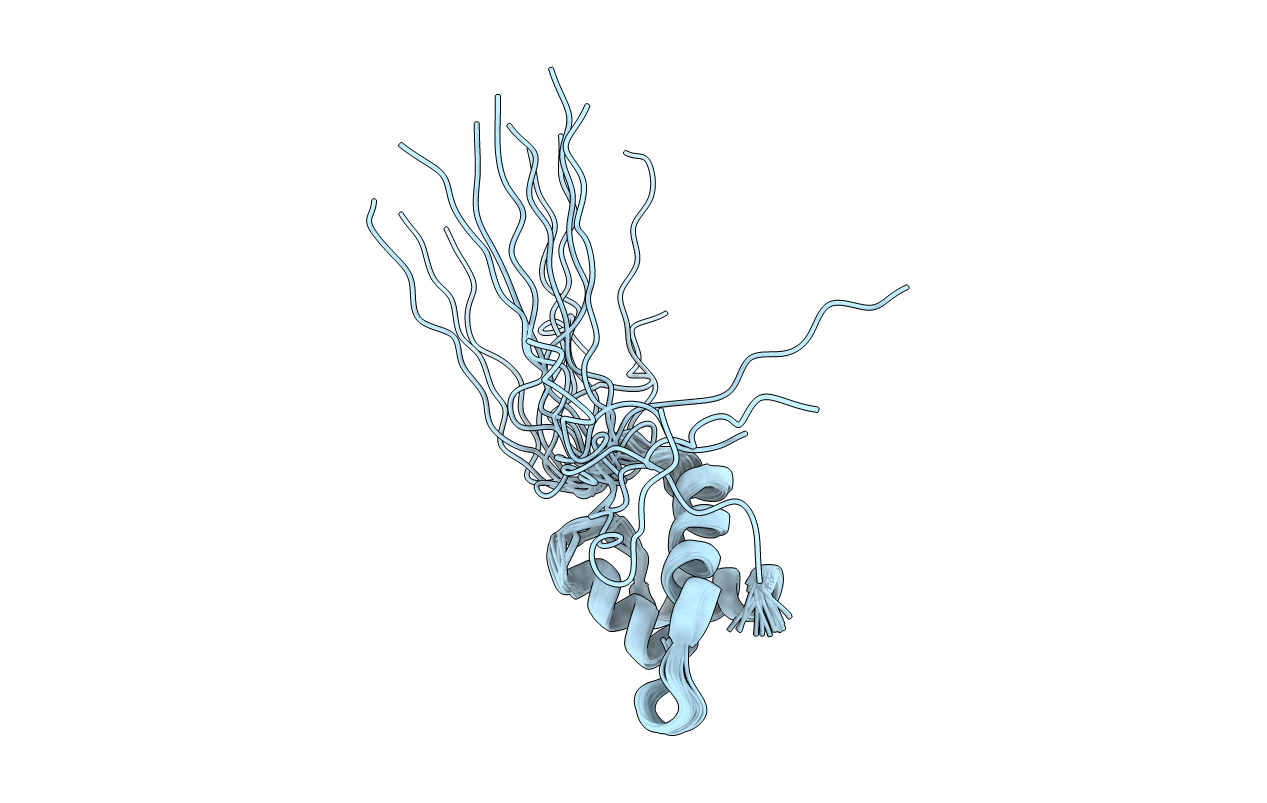
Deposition Date
2005-07-28
Release Date
2005-10-11
Last Version Date
2024-05-22
Entry Detail
PDB ID:
2AHQ
Keywords:
Title:
Solution Structure of the C-terminal RpoN Domain of Sigma-54 from Aquifex aeolicus
Biological Source:
Source Organism(s):
Aquifex aeolicus (Taxon ID: 63363)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
150
Conformers Submitted:
20
Selection Criteria:
target function