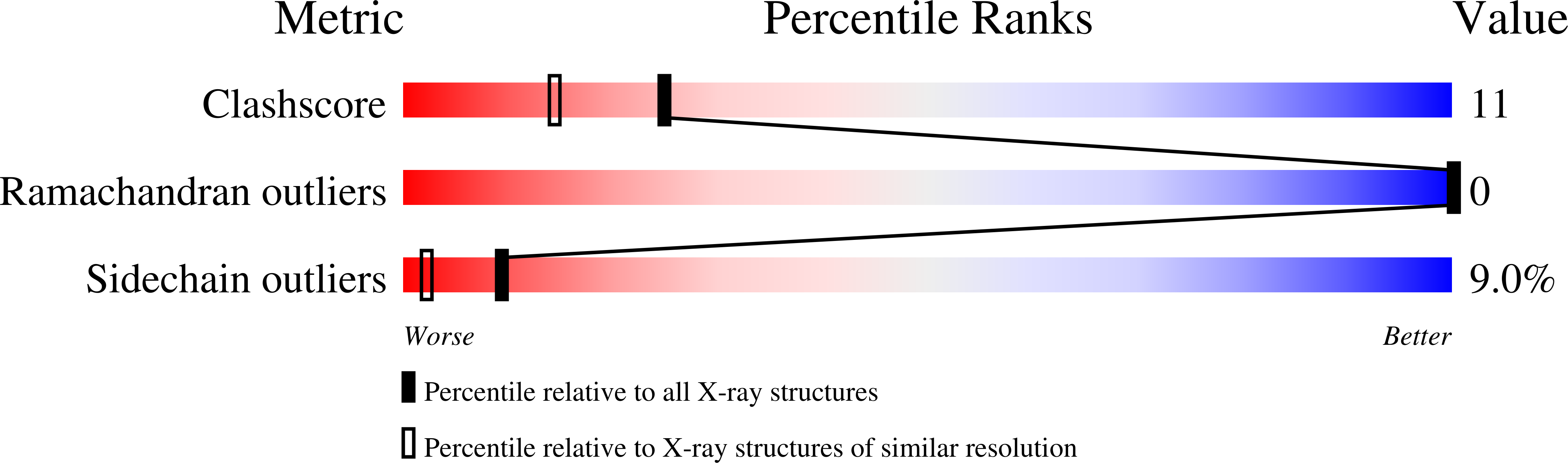Deposition Date
1999-08-17
Release Date
2000-02-06
Last Version Date
2024-11-20
Entry Detail
PDB ID:
1QKZ
Keywords:
Title:
Fab fragment (MN14C11.6) in complex with a peptide antigen derived from Neisseria meningitidis P1.7 serosubtype antigen and domain II from Streptococcal protein G
Biological Source:
Source Organism(s):
STREPTOCOCCUS SP. (Taxon ID: 1320)
NEISSERIA MENINGITIDIS (Taxon ID: 487)
MUS MUSCULUS (Taxon ID: 10090)
NEISSERIA MENINGITIDIS (Taxon ID: 487)
MUS MUSCULUS (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
1.95 Å
R-Value Free:
0.26
R-Value Work:
0.20
R-Value Observed:
0.20
Space Group:
P 1 21 1