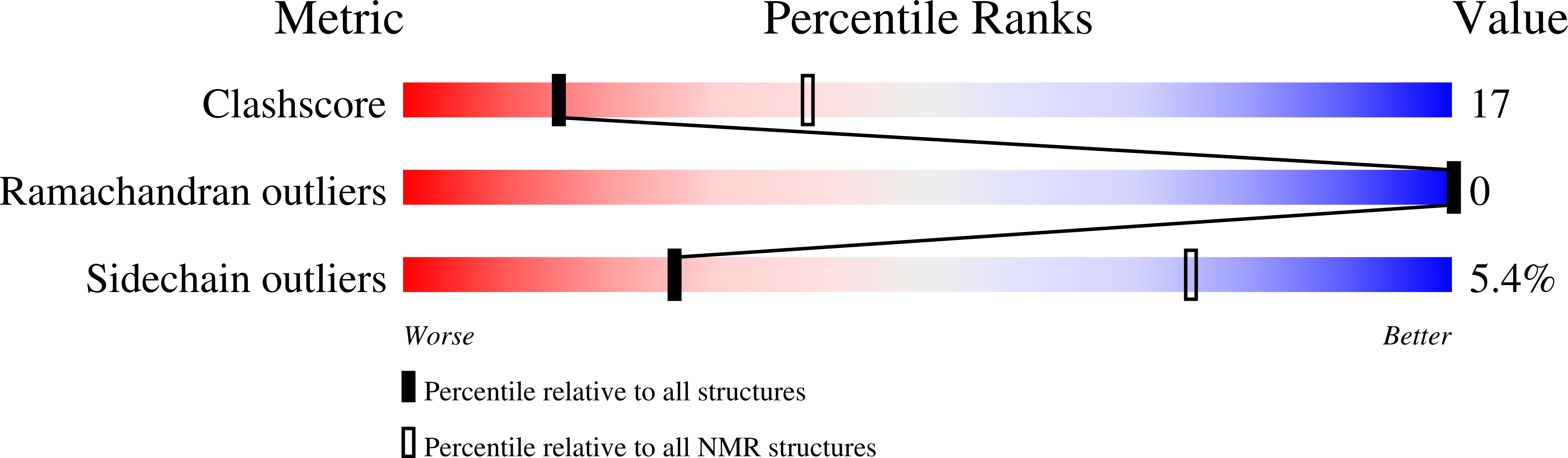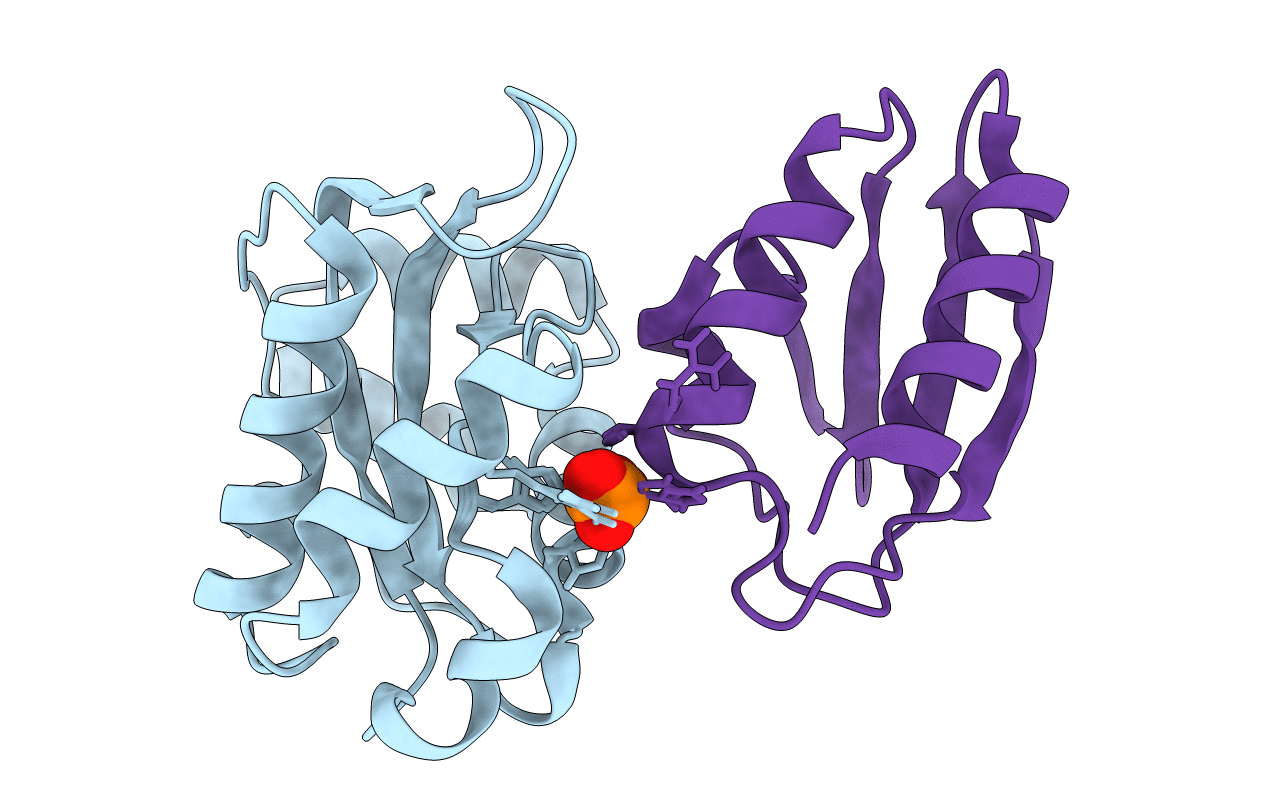
Deposition Date
2002-08-14
Release Date
2002-11-13
Last Version Date
2024-05-22
Entry Detail
PDB ID:
1J6T
Keywords:
Title:
COMPLEX OF ENZYME IIAMTL AND THE HISTIDINE-CONTAINING PHOSPHOCARRIER PROTEIN HPR FROM ESCHERICHIA COLI NMR, RESTRAINED REGULARIZED MEAN STRUCTURE
Biological Source:
Source Organism(s):
Escherichia coli (Taxon ID: 562)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
200
Conformers Submitted:
3
Selection Criteria:
REGULARIZED MEAN STRUCTURES