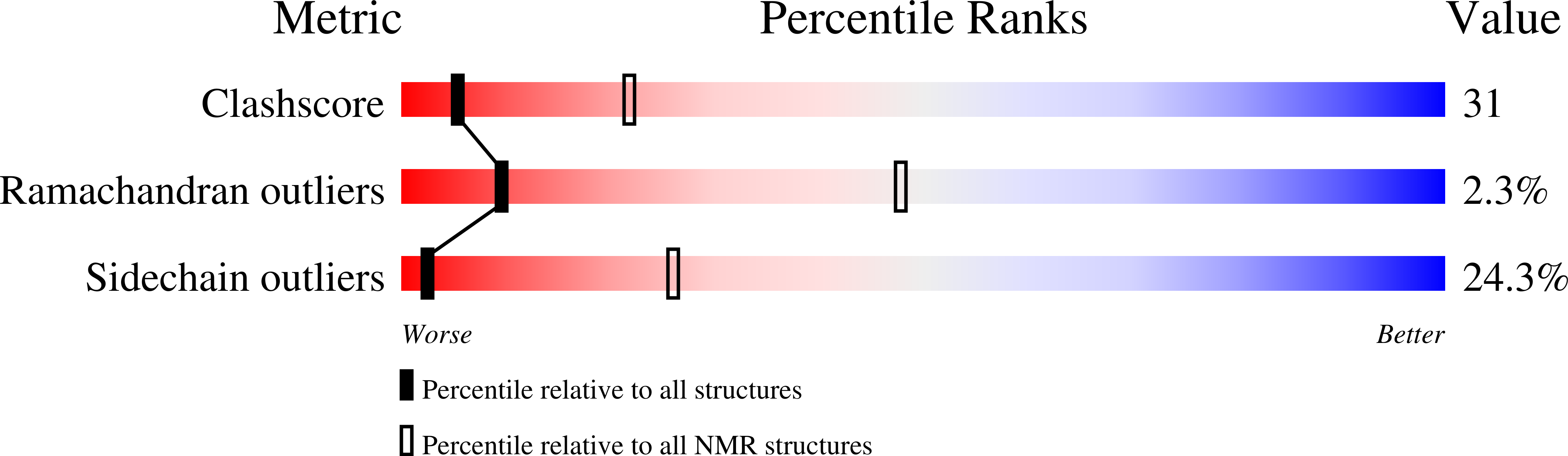
Deposition Date
1994-08-12
Release Date
1995-01-26
Last Version Date
2024-05-22
Entry Detail
PDB ID:
1GBR
Keywords:
Title:
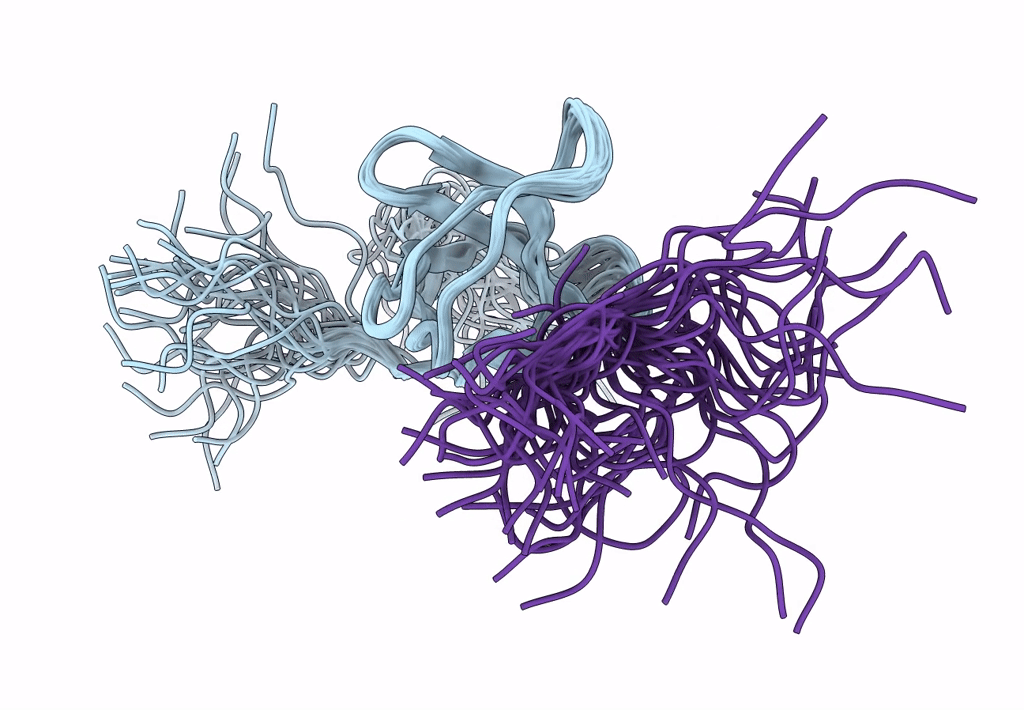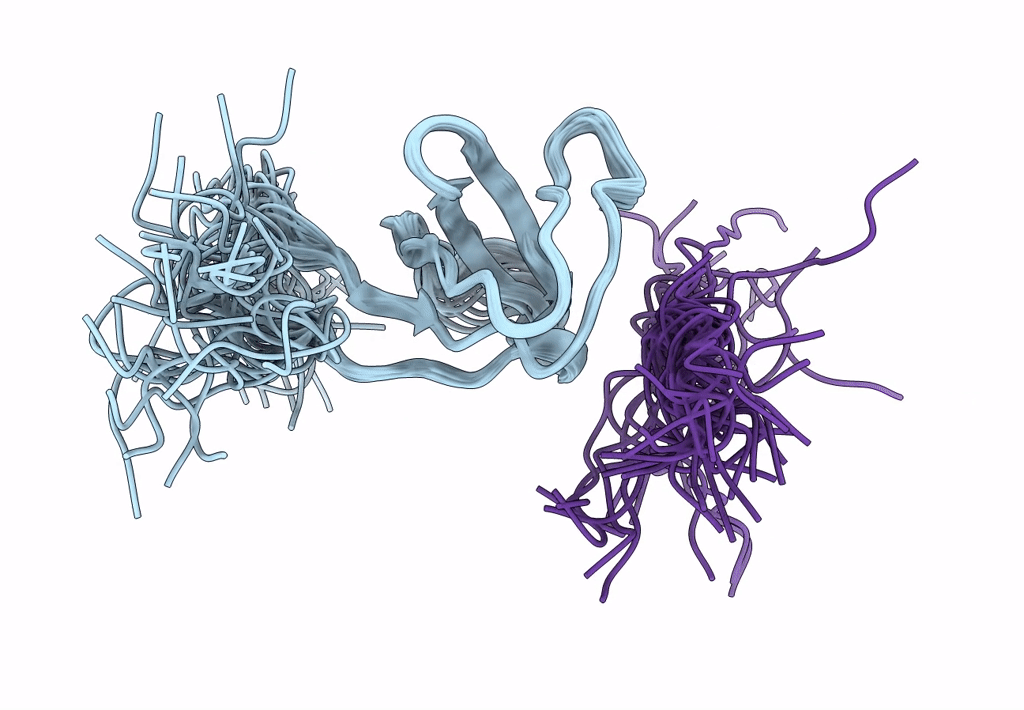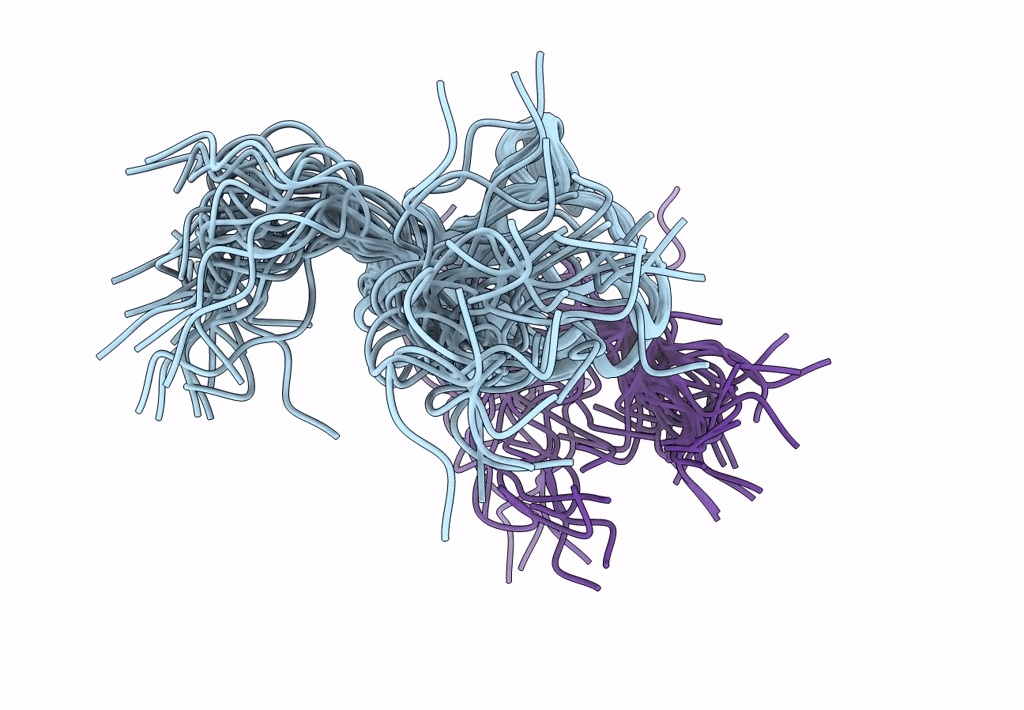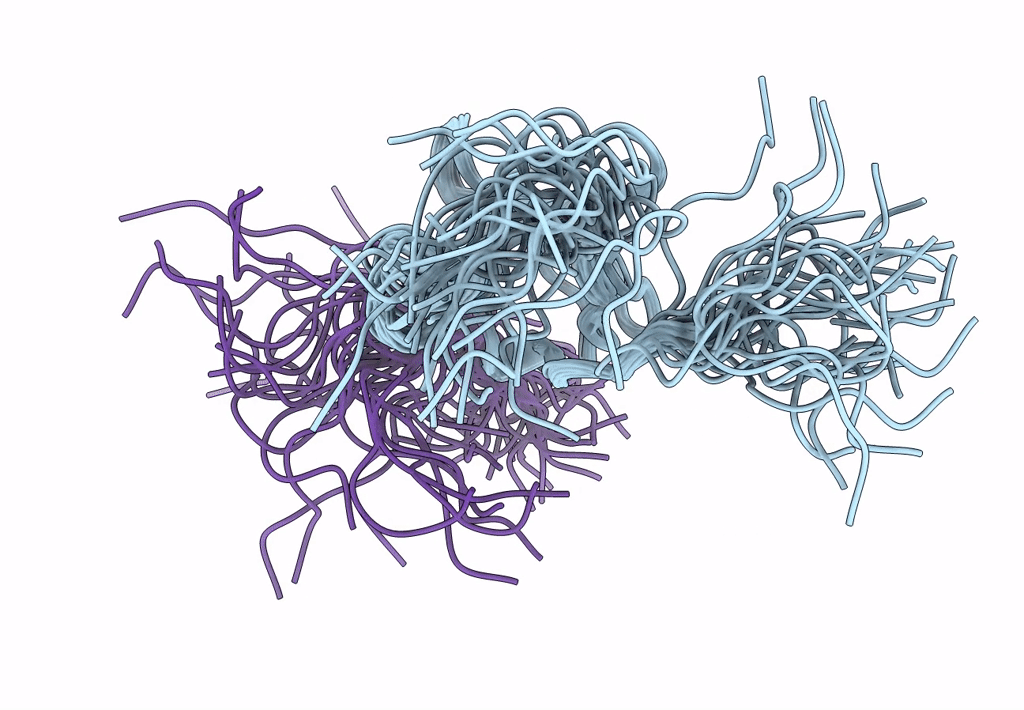
ORIENTATION OF PEPTIDE FRAGMENTS FROM SOS PROTEINS BOUND TO THE N-TERMINAL SH3 DOMAIN OF GRB2 DETERMINED BY NMR SPECTROSCOPY
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
Conformers Submitted:
29