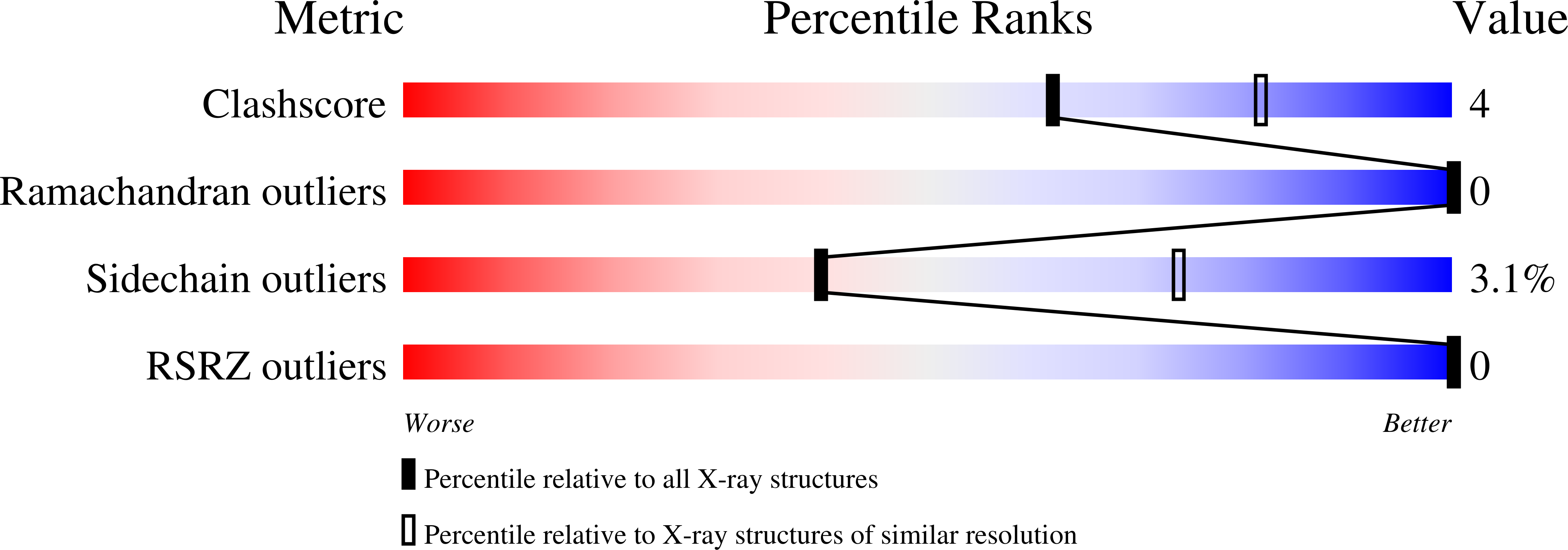
Deposition Date
1999-04-21
Release Date
1999-04-30
Last Version Date
2023-08-09
Entry Detail
Biological Source:
Source Organism(s):
Pseudomonas fluorescens (Taxon ID: 294)
Expression System(s):
Method Details: