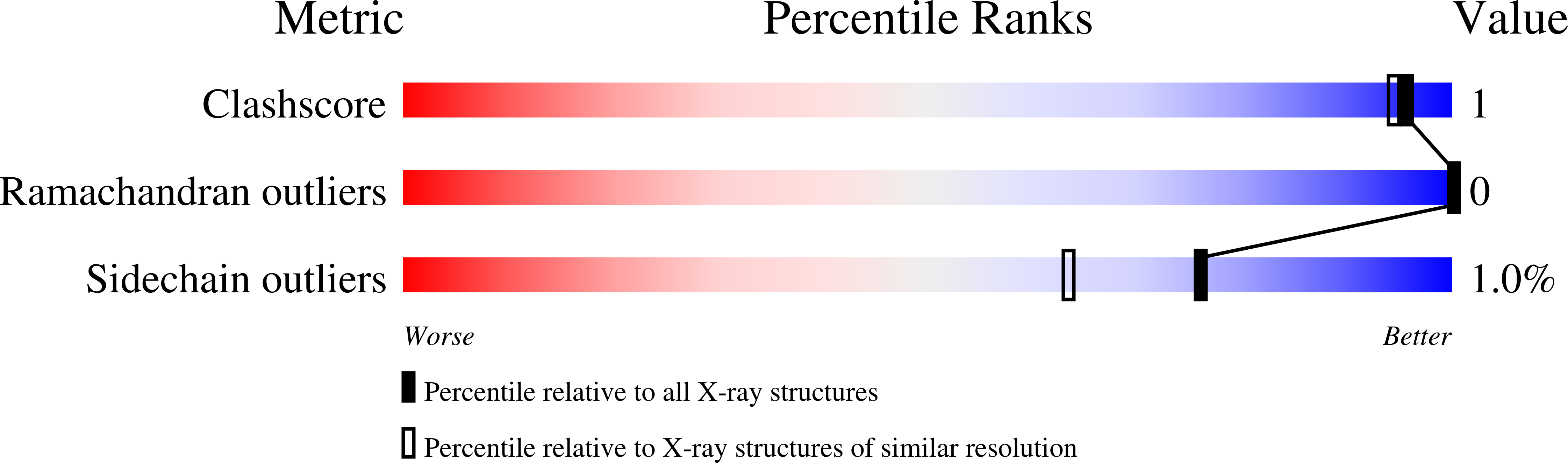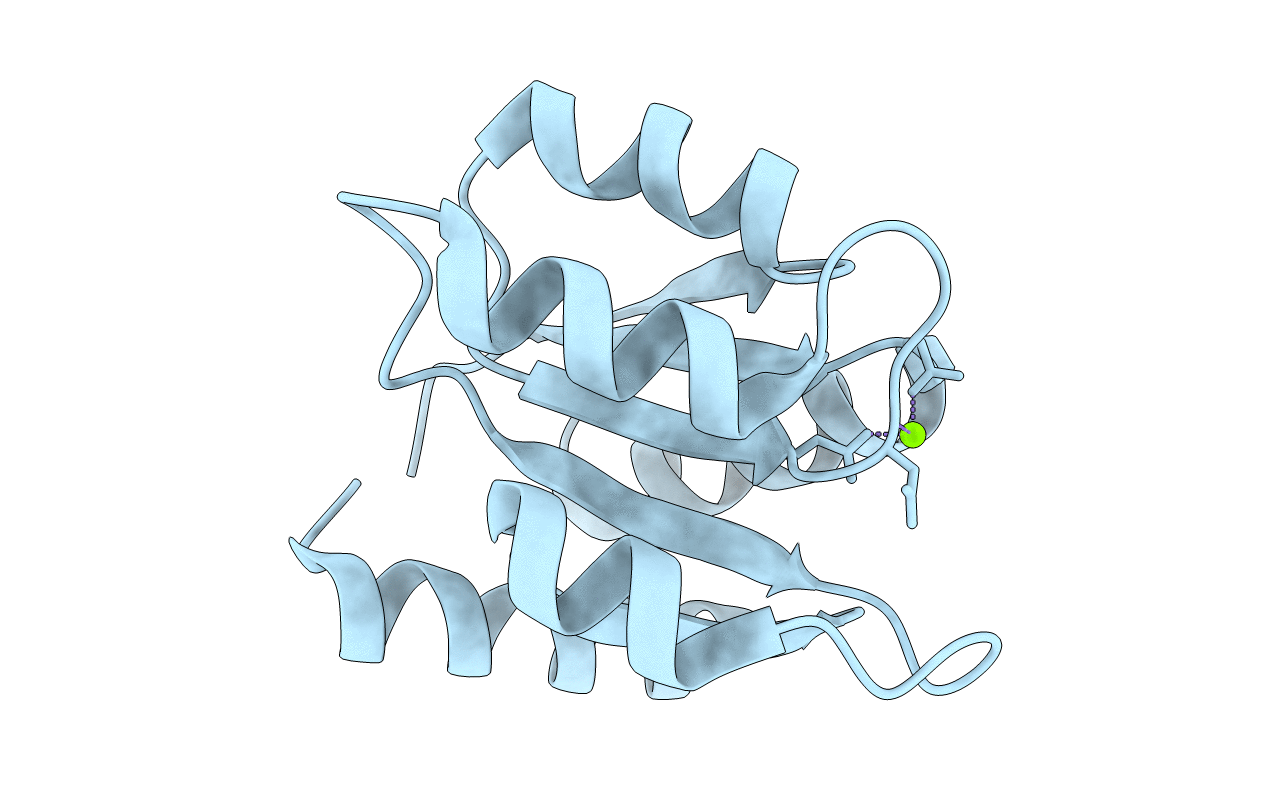
Deposition Date
1994-04-20
Release Date
1994-07-31
Last Version Date
2024-02-07
Entry Detail
PDB ID:
1CHN
Keywords:
Title:
MAGNESIUM BINDING TO THE BACTERIAL CHEMOTAXIS PROTEIN CHEY RESULTS IN LARGE CONFORMATIONAL CHANGES INVOLVING ITS FUNCTIONAL SURFACE
Biological Source:
Source Organism(s):
Escherichia coli (Taxon ID: 562)
Method Details:
Experimental Method:
Resolution:
1.76 Å
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 1 21 1