Deposition Date
2025-12-19
Release Date
2026-01-21
Last Version Date
2026-02-18
Entry Detail
PDB ID:
9TQB
Keywords:
Title:
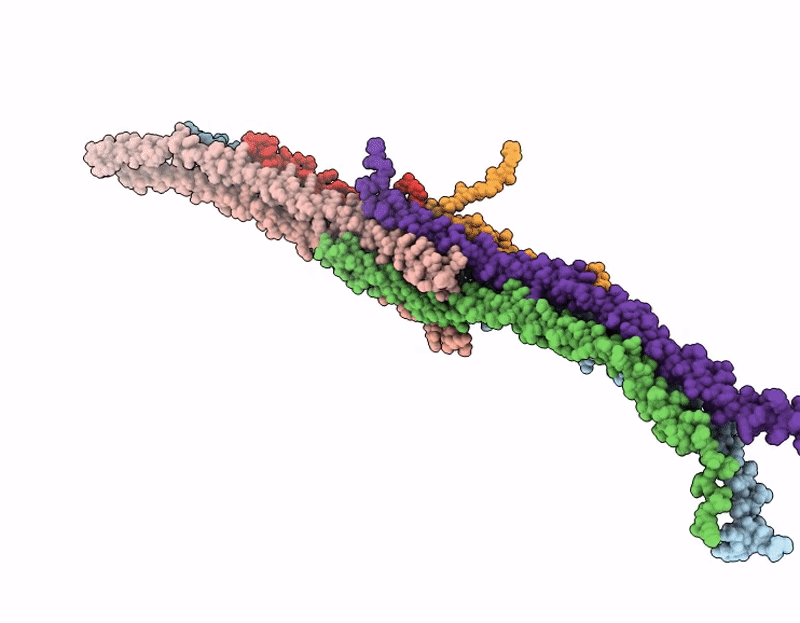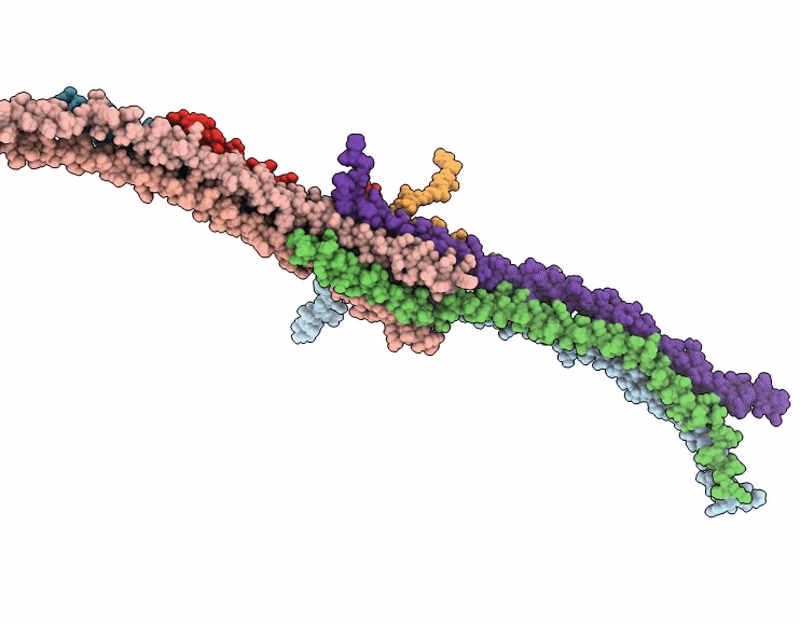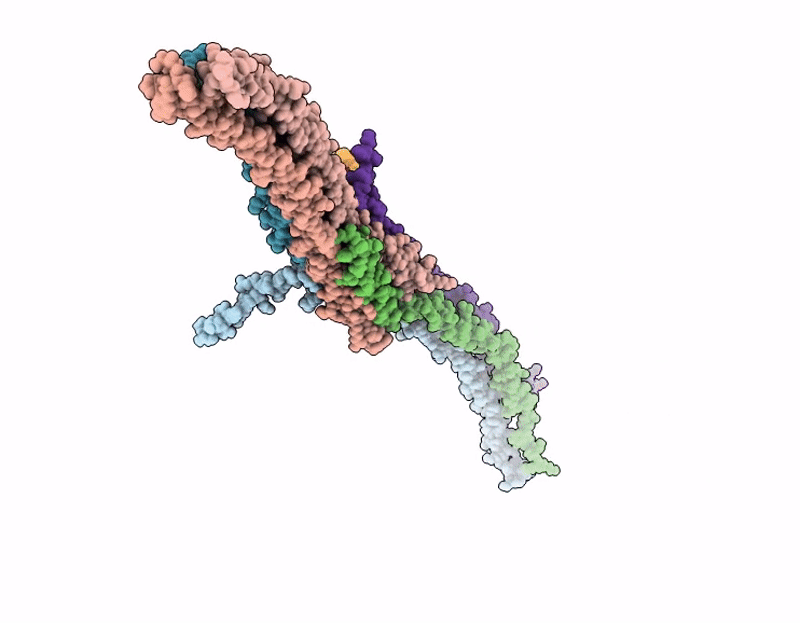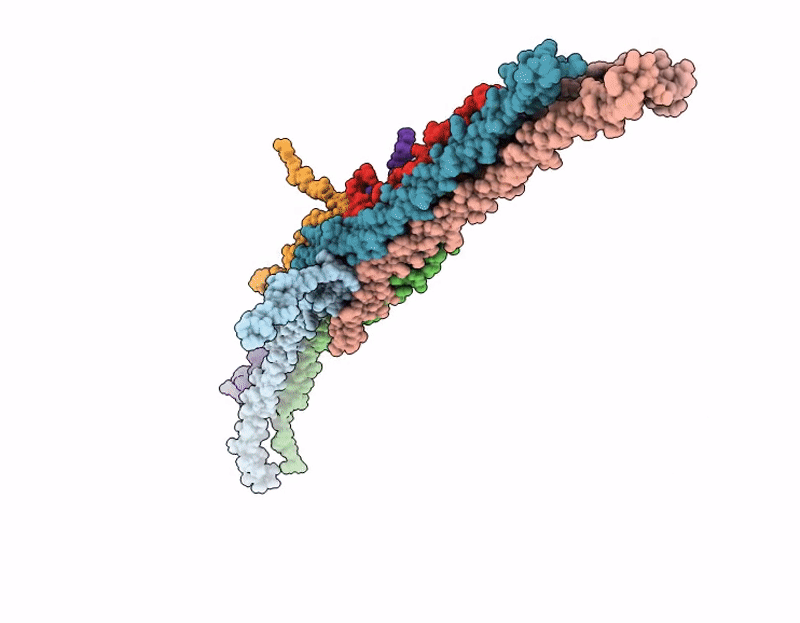
Octameric C. elegans BORC, containing BORCS5, BORCS6, BORCS7, BORCS8, KXD1 and the shared BORC and BLoC-1 subunits, BLOC1S1, BLOC1S2 and Snapin
Biological Source:
Source Organism(s):
Caenorhabditis elegans (Taxon ID: 6239)
Caenorhabditis (Taxon ID: 6237)
Caenorhabditis (Taxon ID: 6237)
Expression System(s):
Method Details:
Experimental Method:
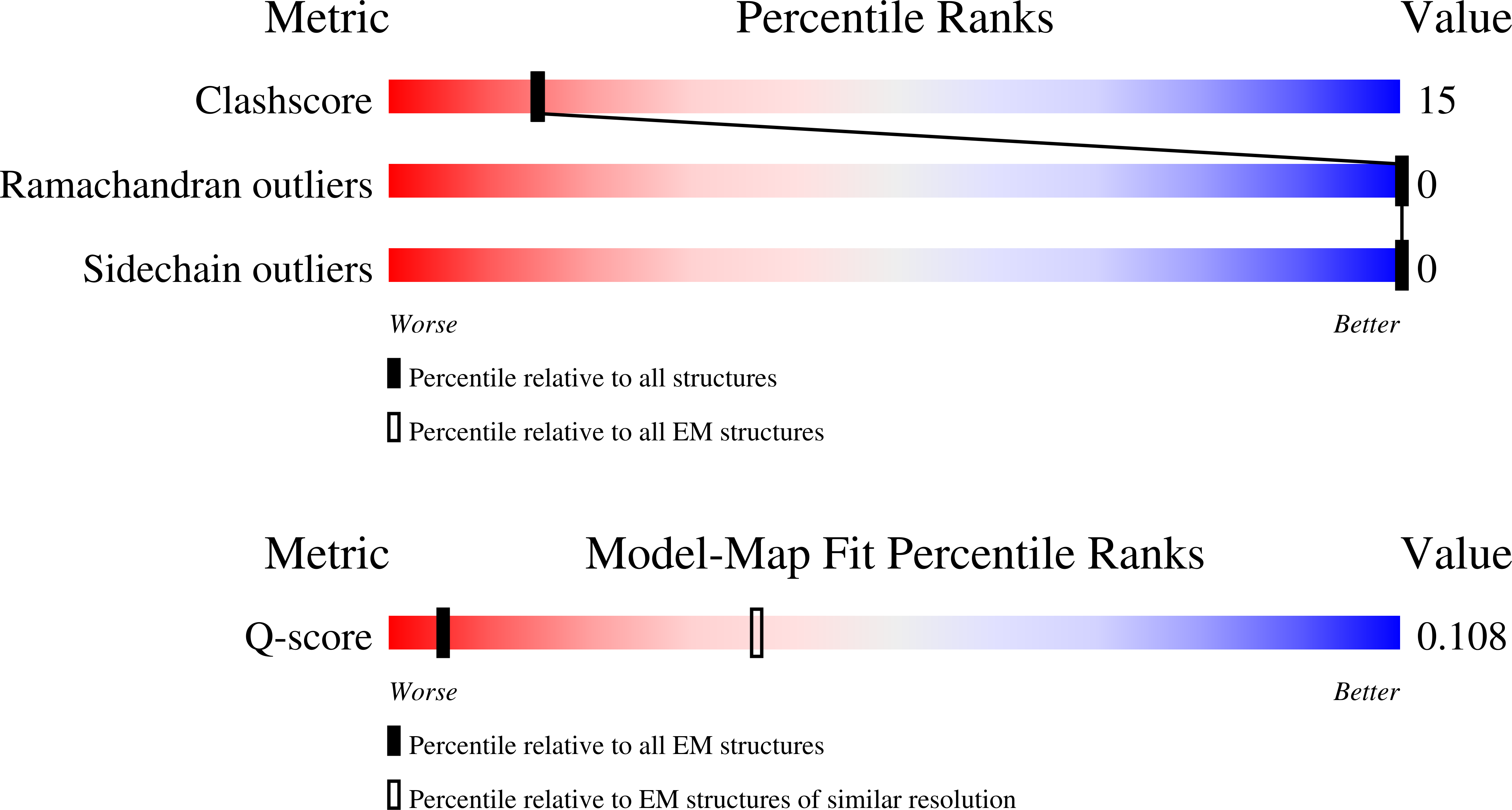
Resolution:
7.80 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE