Deposition Date
2025-03-09
Release Date
2026-01-21
Last Version Date
2026-01-21
Entry Detail
PDB ID:
9QEF
Keywords:
Title:
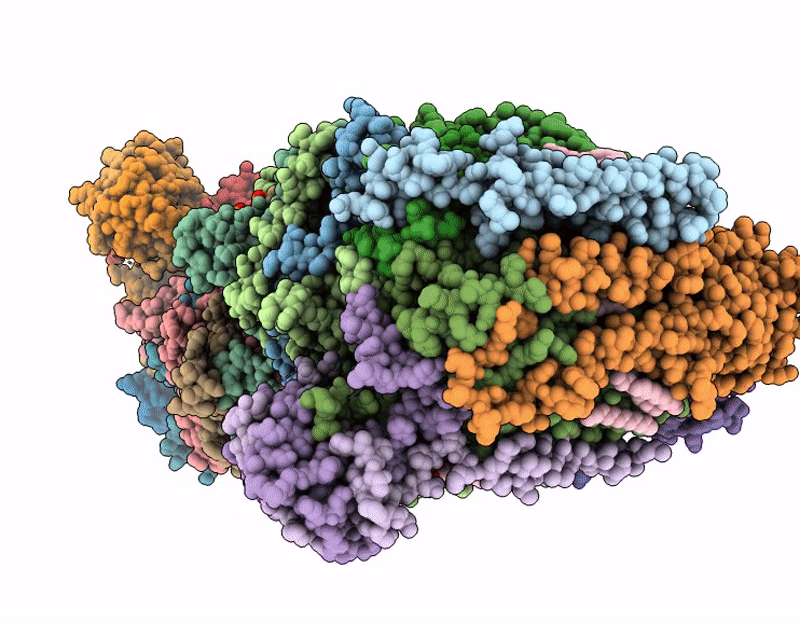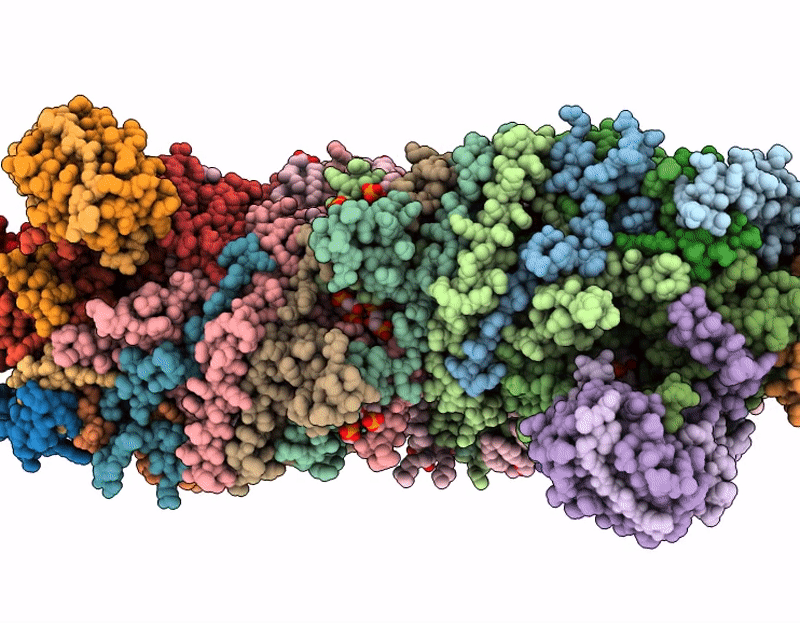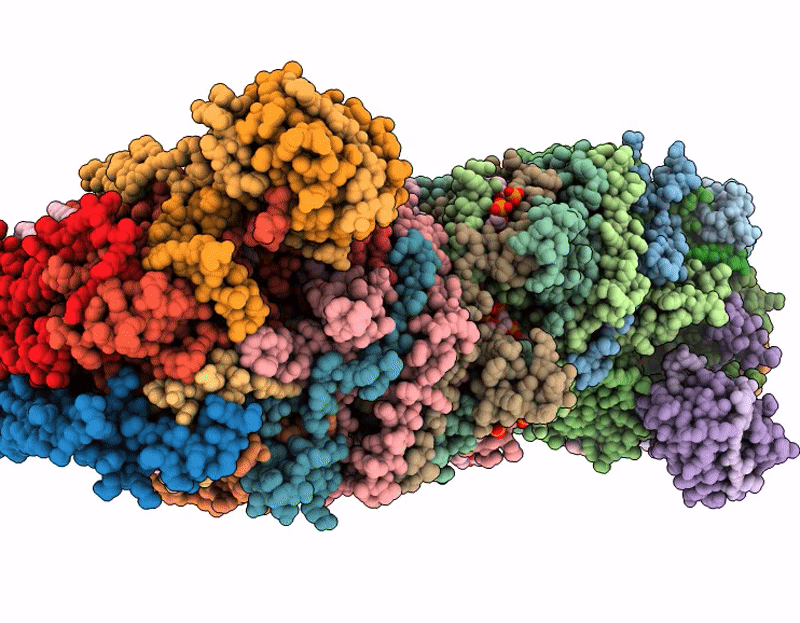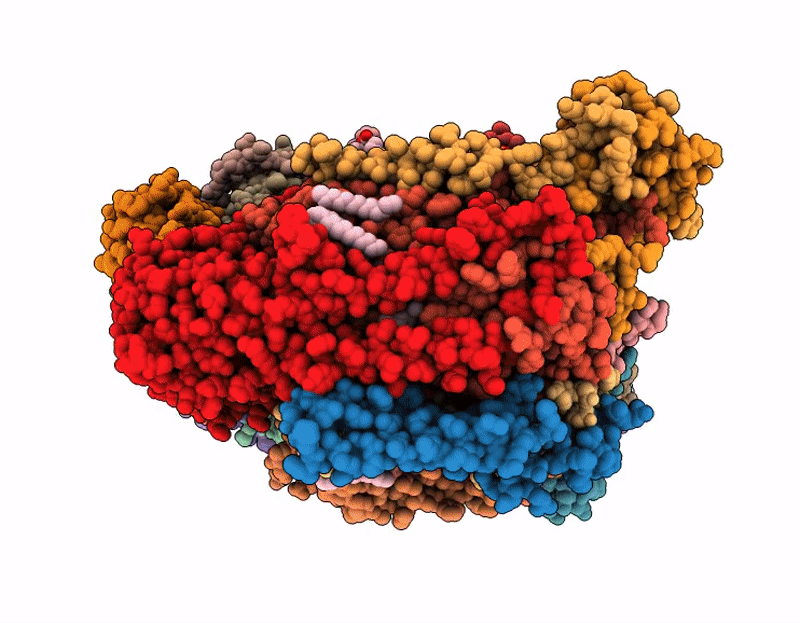
Respiratory supercomplex from Mycobacterium smegmatis with decylubiquinone
Biological Source:
Source Organism(s):
Mycolicibacterium smegmatis (Taxon ID: 1772)
Expression System(s):
Method Details:
Experimental Method:
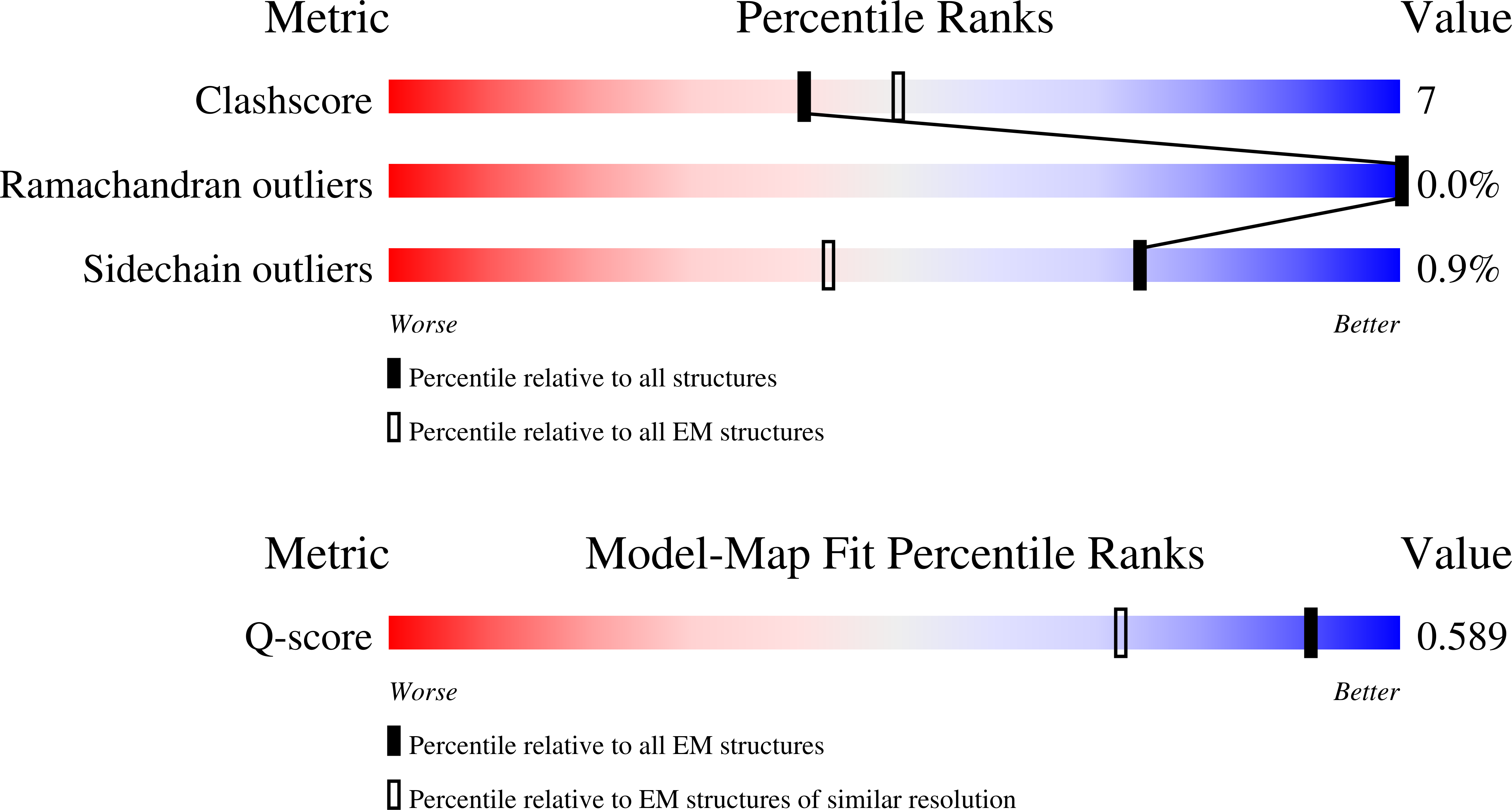
Resolution:
2.50 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE