Deposition Date
2025-05-06
Release Date
2026-01-28
Last Version Date
2026-02-11
Entry Detail
PDB ID:
9OIK
Keywords:
Title:
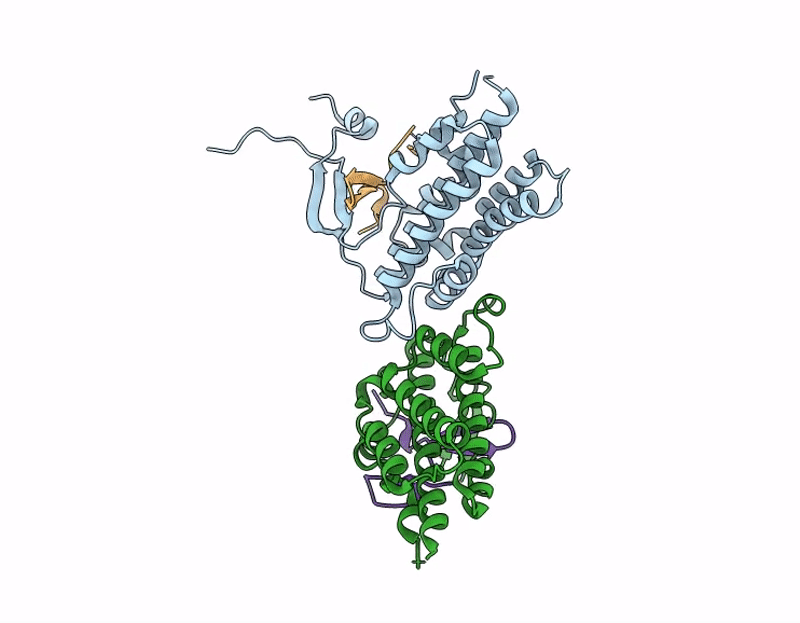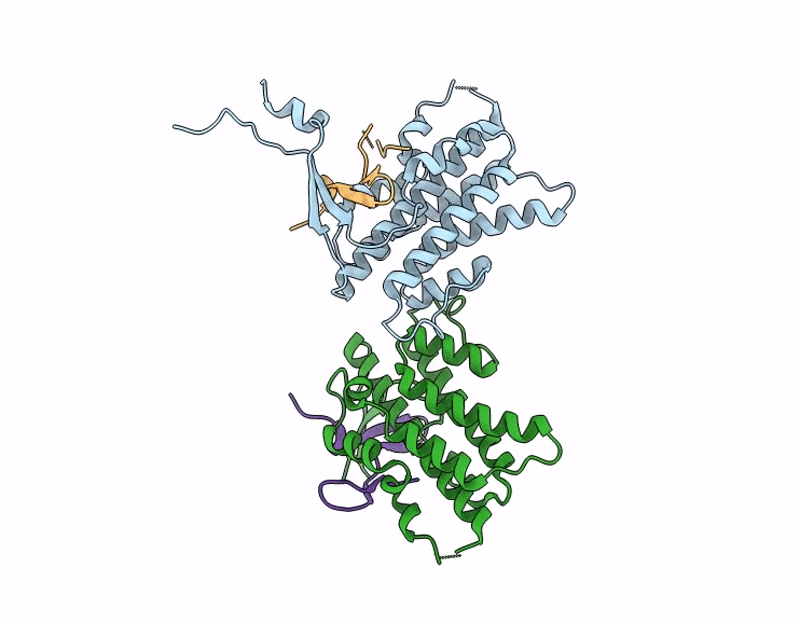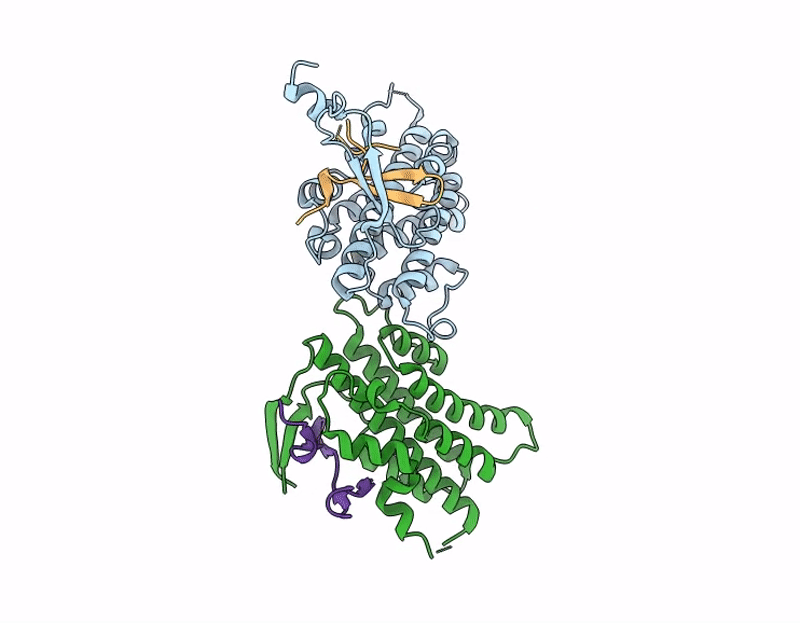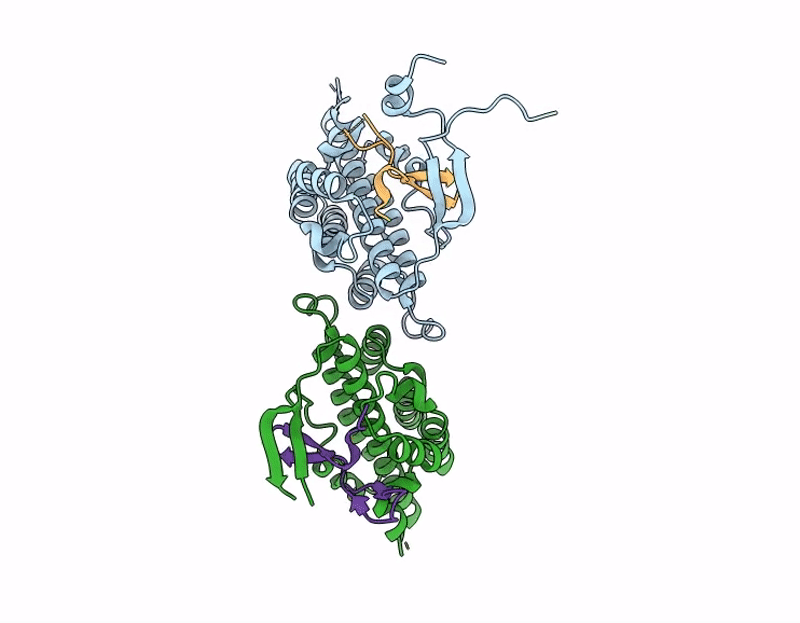
Structure of S. Typhimurium 14028 Gifsy-1 prophage HepS bound to bacteriophage lambda J Tail Tip
Biological Source:
Source Organism(s):
Expression System(s):
Method Details:
Experimental Method:
Resolution:
1.86 Å
R-Value Free:
0.20
R-Value Work:
0.18
R-Value Observed:
0.18
Space Group:
C 1 2 1


