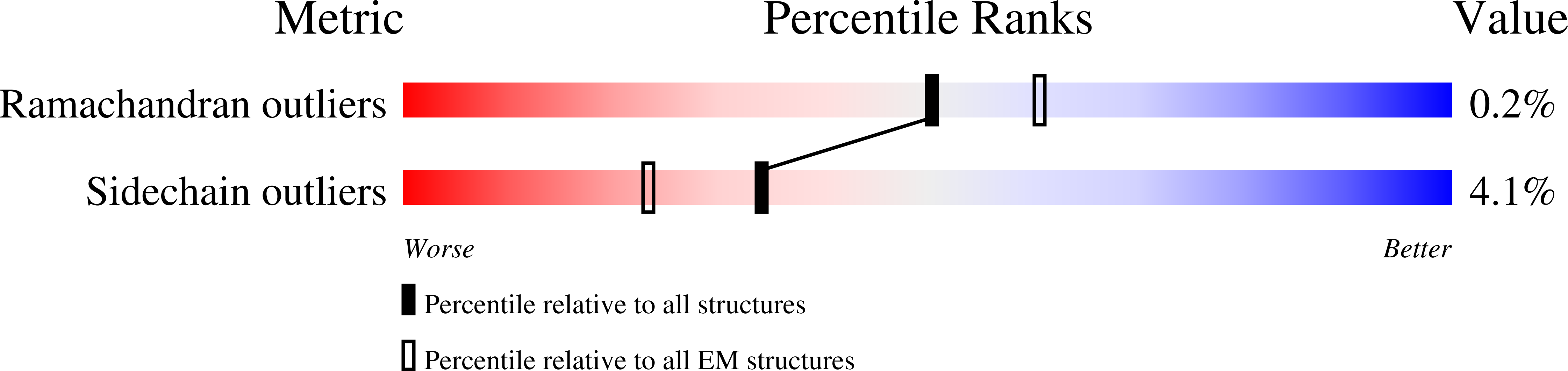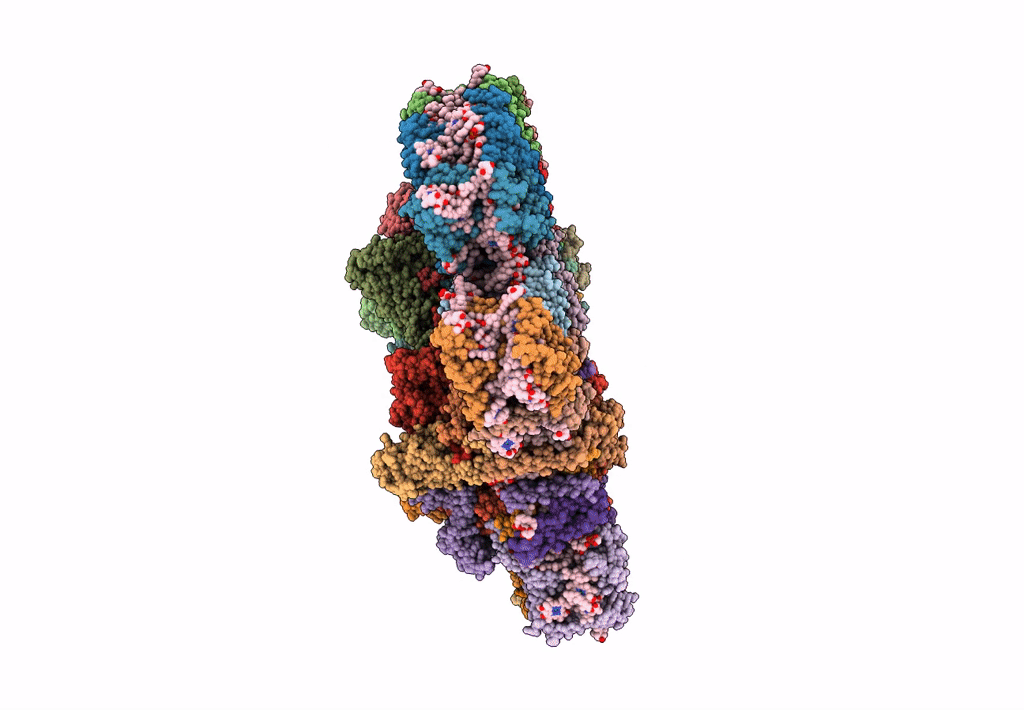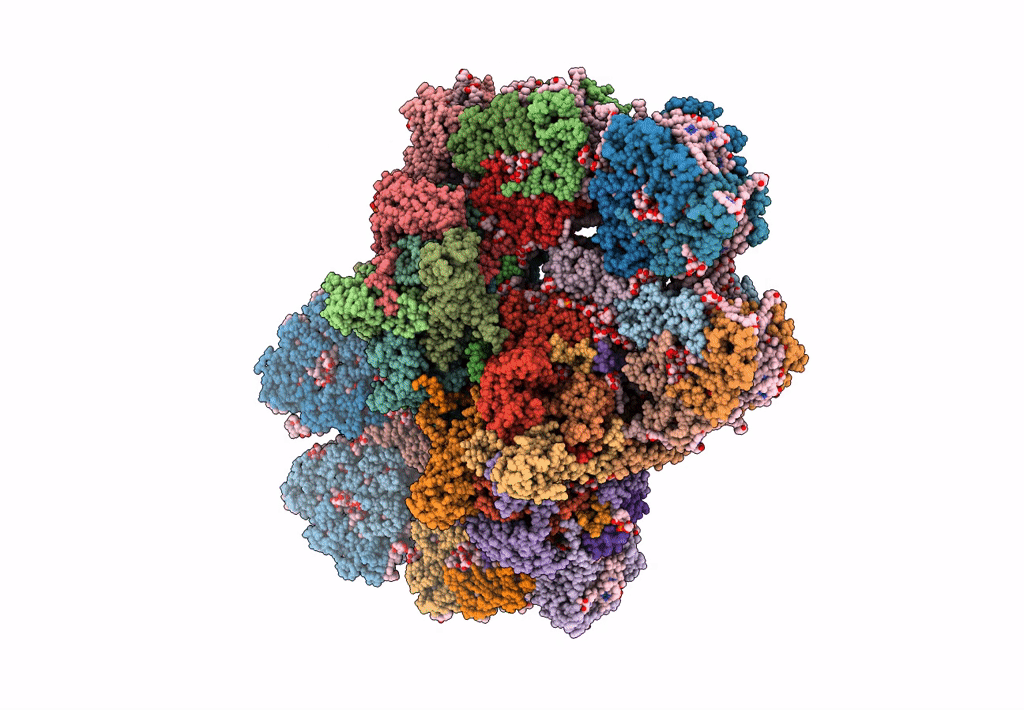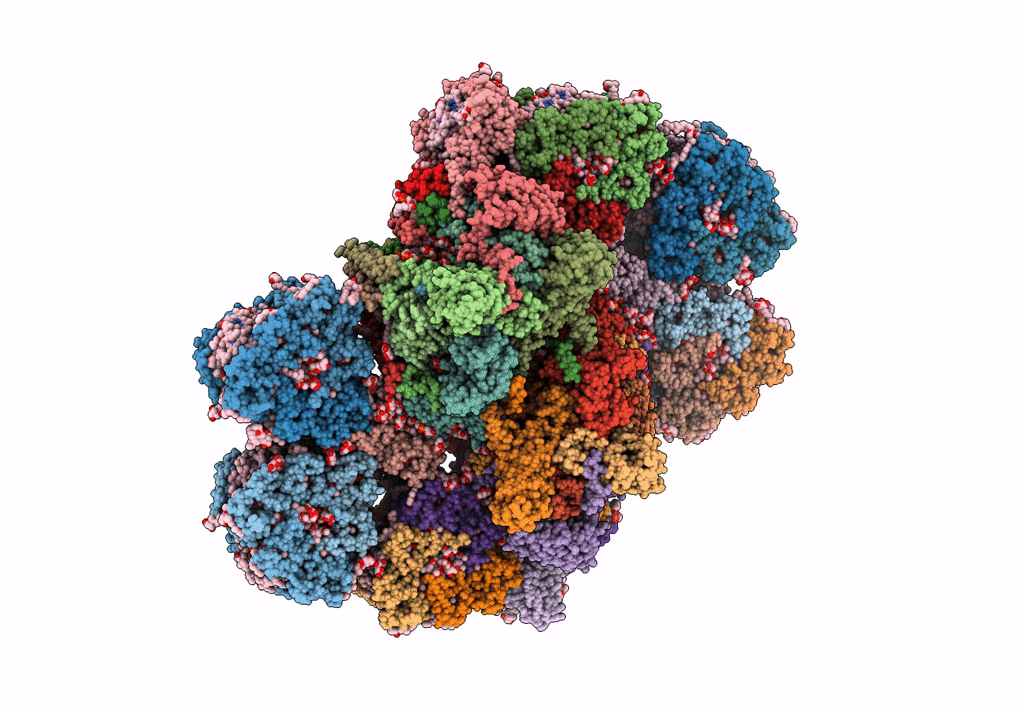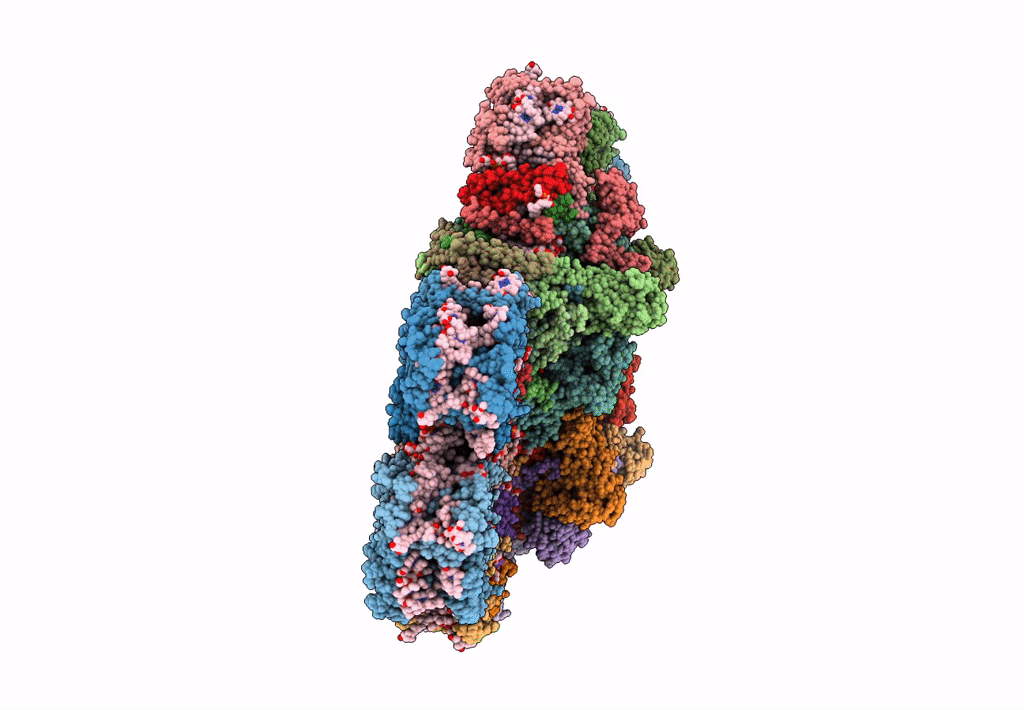
Deposition Date
2022-10-18
Release Date
2023-08-09
Last Version Date
2023-08-23
Entry Detail
PDB ID:
8BD3
Keywords:
Title:
Cryo-EM structure of the Photosystem II - LHCII supercomplex from Chlorella ohadi
Biological Source:
Source Organism(s):
Chlorella ohadii (Taxon ID: 2649997)
Method Details:
Experimental Method:
Resolution:
2.73 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE