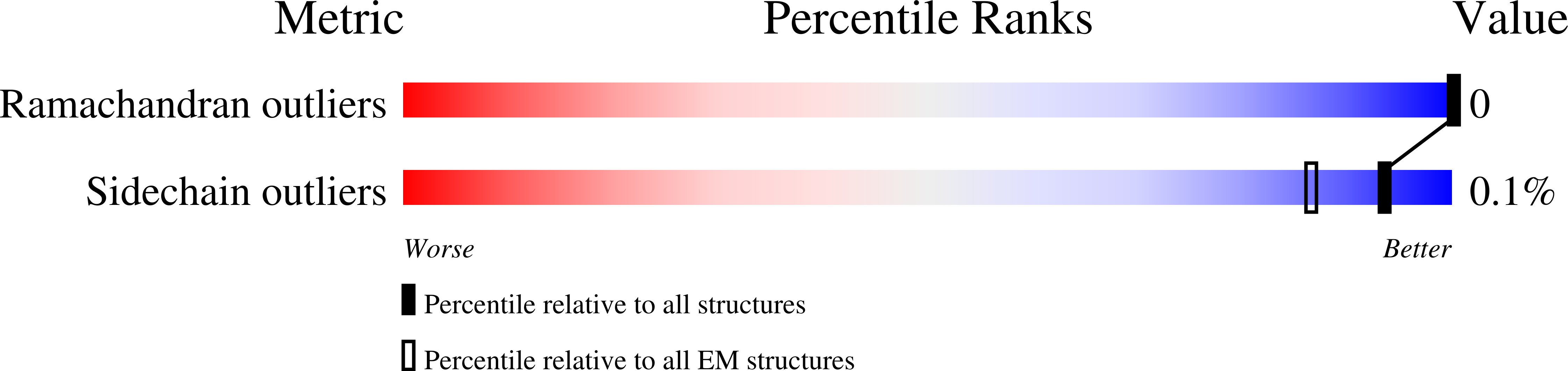
Deposition Date
2021-09-22
Release Date
2022-04-06
Last Version Date
2026-03-04
Entry Detail
PDB ID:
7PSA
Keywords:
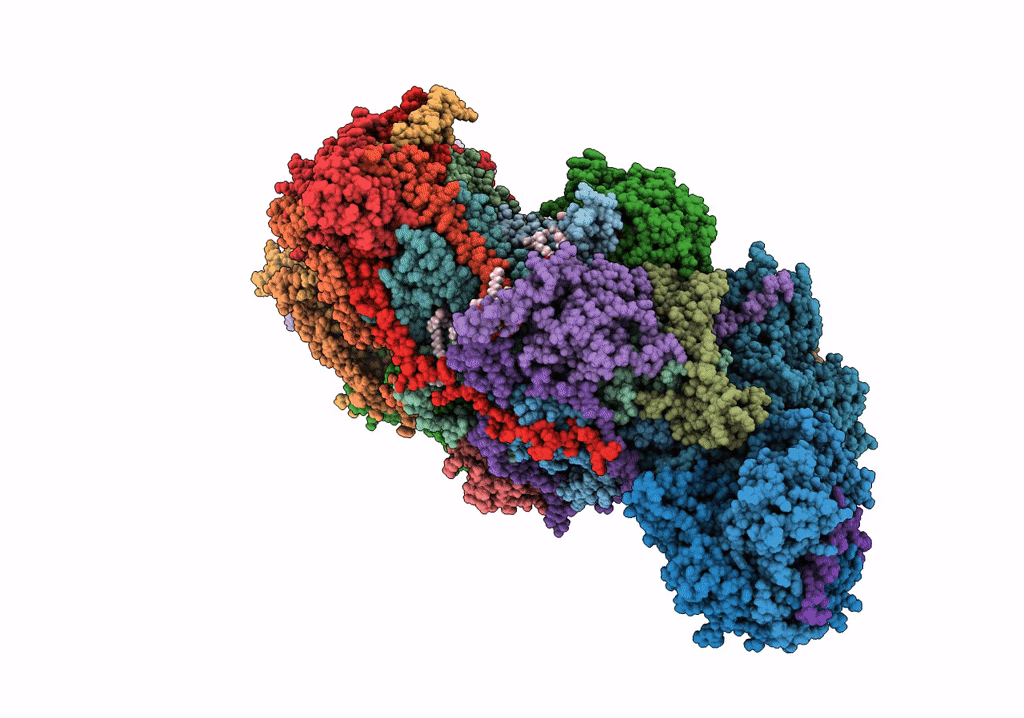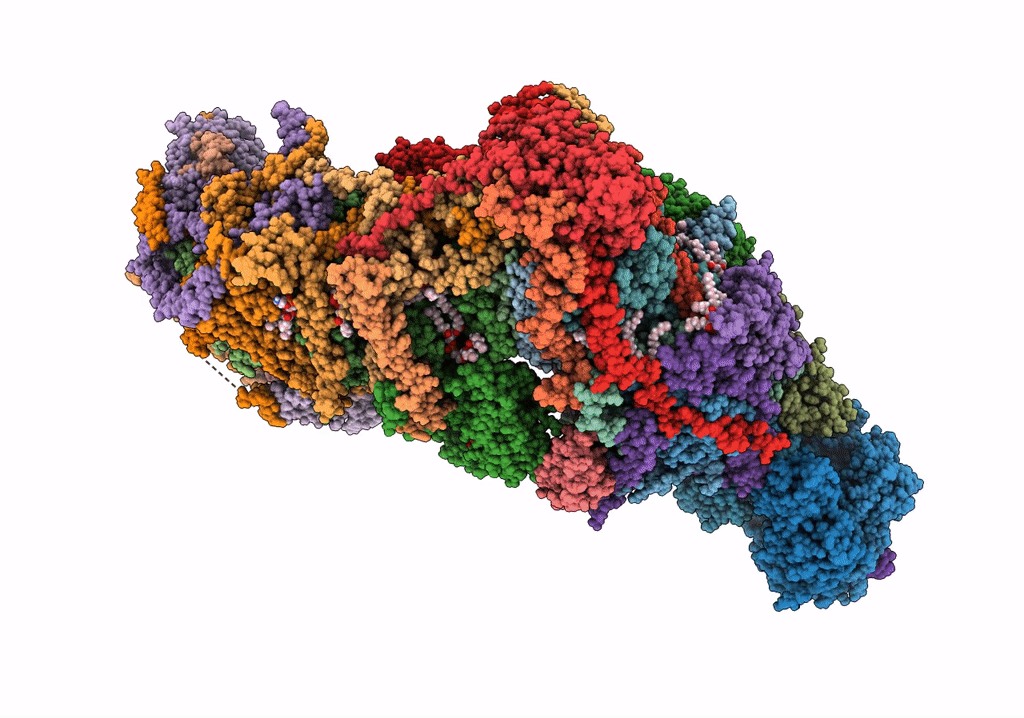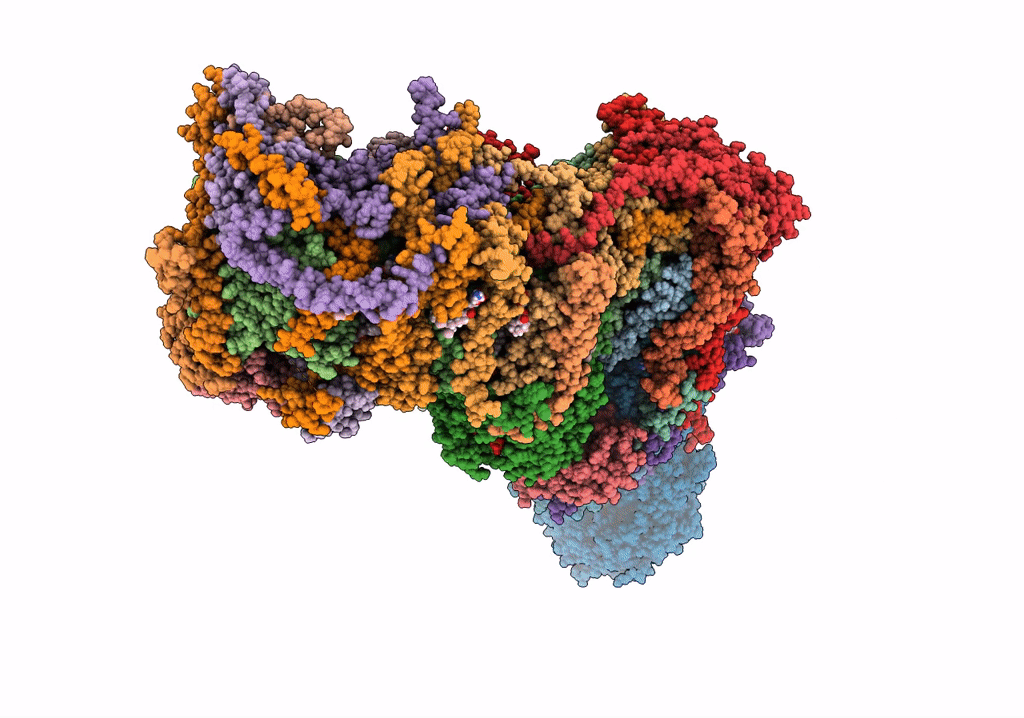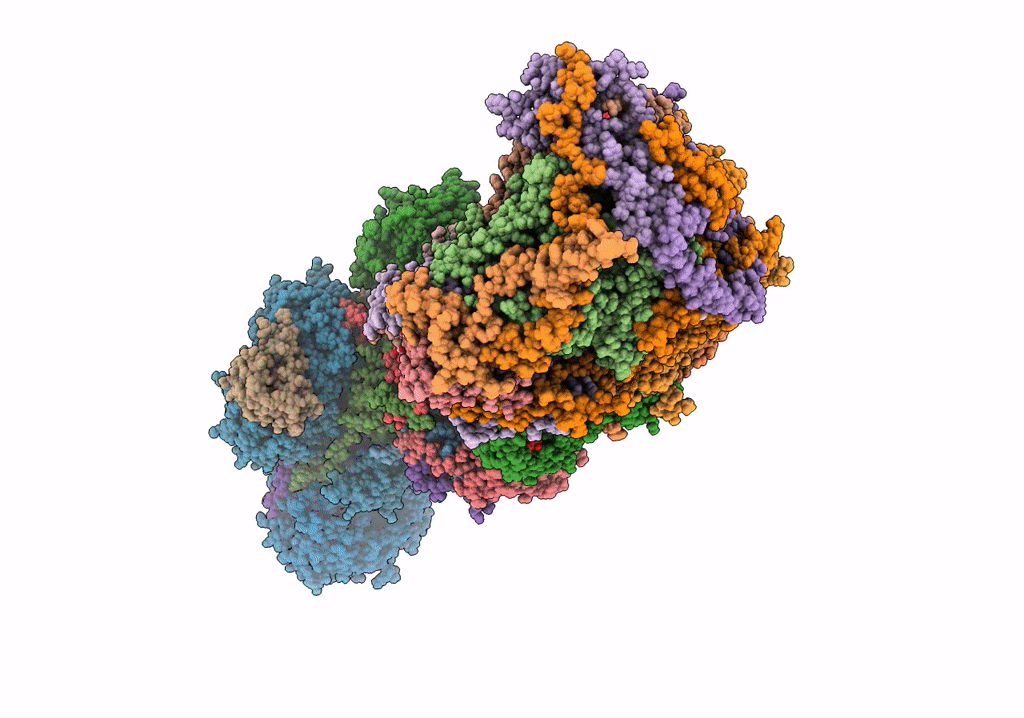
Title:
The acetogenin-bound complex I of Mus musculus resolved to 3.4 angstroms
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Method Details:
Experimental Method:
Resolution:
3.40 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE