Deposition Date
2017-03-20
Release Date
2017-06-21
Last Version Date
2024-11-20
Entry Detail
PDB ID:
5V7J
Keywords:
Title:
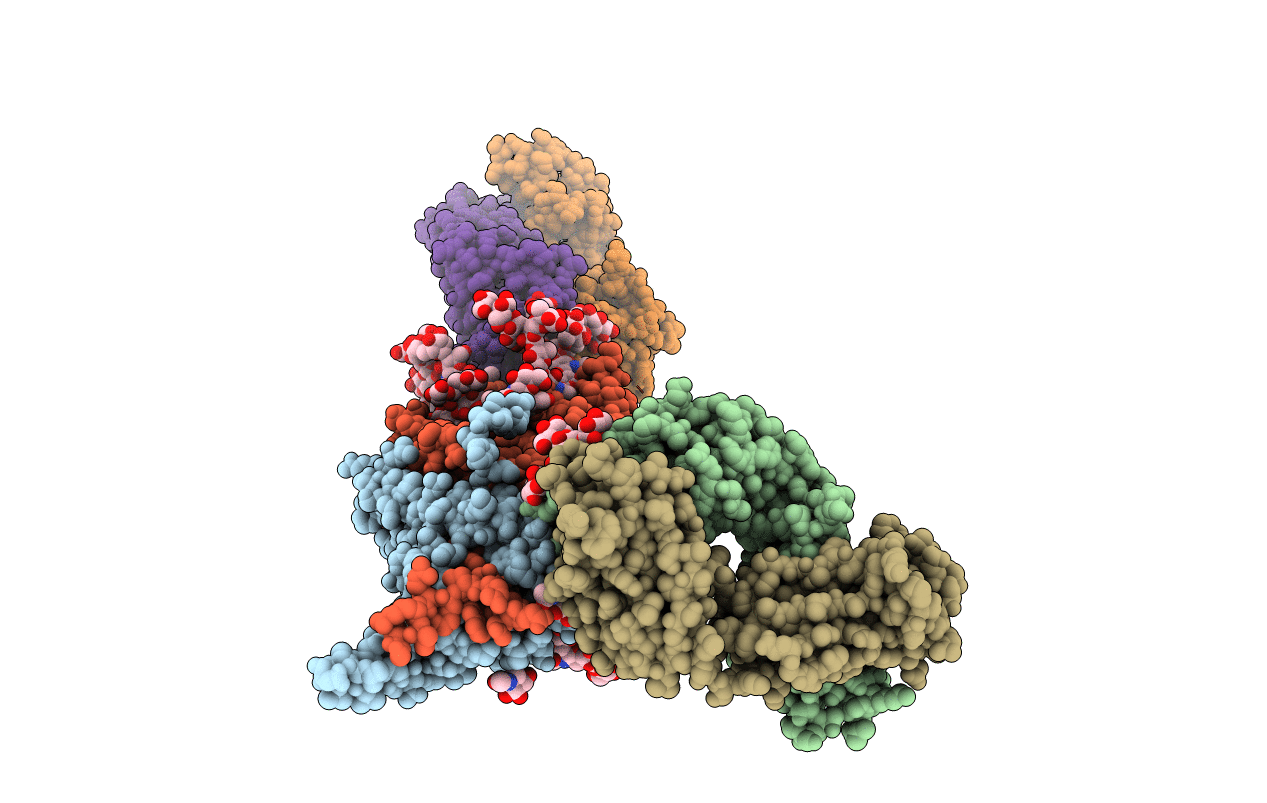
Crystal Structure at 3.7 A Resolution of Glycosylated HIV-1 Clade A BG505 SOSIP.664 Prefusion Env Trimer with Four Glycans (N197, N276, N362, and N462) removed in Complex with Neutralizing Antibodies 3H+109L and 35O22.
Biological Source:
Source Organism(s):
Human immunodeficiency virus 1 (Taxon ID: 11676)
Homo sapiens (Taxon ID: 9606)
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
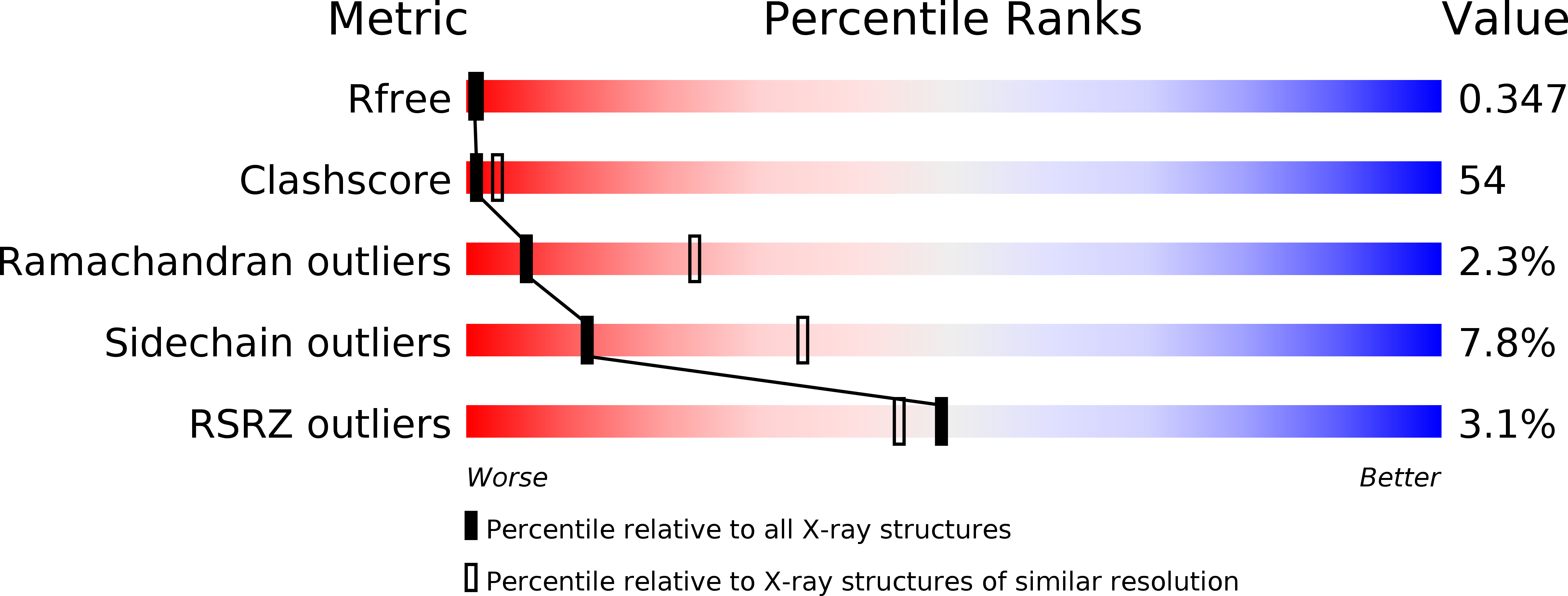
Resolution:
2.91 Å
R-Value Free:
0.33
R-Value Work:
0.31
R-Value Observed:
0.32
Space Group:
P 63